Shellfish chemistry combined with polymer to create new biodegradable adhesive
A new type of adhesive that combines the bonding chemistry of shellfish with a bio-based polymer has been shown to perform as well as commercially available products and can be easily degraded, representing a potential non-toxic alternative.
"Adhesives releasing toxins including carcinogenic formaldehyde are almost everywhere in our homes and offices. The plywood in our walls, the chairs we sit on, and the carpet beneath our feet are all off-gassing reactive chemicals" said Jonathan Wilker, a professor of chemistry and materials engineering at Purdue University. "Most of these glues are also permanent, preventing disassembly and recycling of electronics, furniture and automobiles. In order to develop the next generation of advanced adhesives we have turned to biology for inspiration."
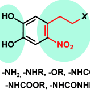
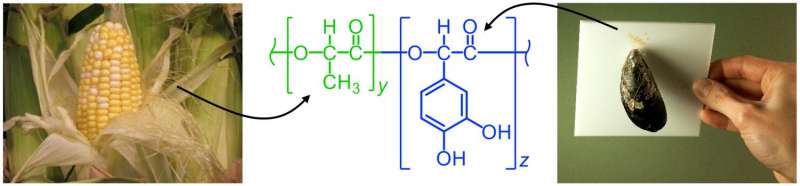
Mussels extend hair-like fibers that attach to surfaces using plaques of adhesive. Proteins in the glue contain the amino acid DOPA, which harbors the chemistry needed to facilitate the "cross-linking" of protein molecules, providing strength and adhesion. Purdue researchers have now combined this bonding chemistry of mussel proteins with a polymer called poly(lactic acid), or PLA, a bio-based polymer that can be derived from corn. The adhesive was created by harnessing the chemistry of compounds called catechols, contained in DOPA.
"We found the adhesive bonding to be appreciable and comparable to several petroleum-based commercial glues," Wilker said.
Findings are detailed in a research paper published online Jan. 4 in the journal Macromolecules and to appear in an upcoming print issue of the journal. The paper was authored by Wilker and graduate students Courtney L. Jenkins and Heather M. Siebert in Purdue's Department of Chemistry. Jenkins is now an assistant professor of chemistry at Ball State University.
"Results presented here show that a promising new adhesive system can be derived from a renewable resource, display high-strength bonding, and easily degrade in a controlled fashion," Wilker said. "Particularly unique was the ability to debond this adhesive under mild conditions."
Early adhesives were made of natural materials such as starch, but have been replaced in recent decades with synthetic glues possessing superior performance. About 9 billion kilograms of glue are now manufactured annually in the United States, with nearly 4 billion kilograms containing formaldehyde.
"The detrimental health and environmental effects of synthetic glues are becoming more of a concern, with alternatives being developed," Wilker said. "Renewable, nontoxic, and removable adhesives are thus in great demand to decrease our exposure to pollutants as well as waste in landfills."
The researchers tested the adhesive by measuring the force needed to pull apart metal and plastic plates bonded together, finding that it compared favorably with various commercial products. Unlike synthetic glues, however, the adhesive can be easily degraded in water.
"This new system may help lead us toward nontoxic materials sourced from nature, capable of being broken down into benign components, and enhanced recyclability of the products all around us," Wilker said.
More information: Integrating Mussel Chemistry into a Bio-Based Polymer to Create Degradable Adhesives, Macromolecules, 2017.
Provided by Purdue University