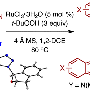Faster, cheaper methods to synthesize compounds used in drug discovery

Chemists at The University of Texas at Arlington are developing new methods to synthesize groups of chemical compounds to provide faster, less expensive routes to produce the compounds for subsequent use in medical drug discovery and development.
The project focuses on compounds containing carbon-silicon bonds, which are called organosilanes or organosilanols. These molecules are in increasingly high demand for use in a variety of applications, including pharmaceutical and other medical-related activities. They are also widely used in organic chemistry, particularly as protecting groups, reducing agents and synthetic intermediates.
"The goal of our research is to develop novel compounds from readily accessible, simple chemical building blocks," said Junha Jeon, UTA assistant professor of chemistry and biochemistry and leader of the project. "The synthetic methods developed in this research will provide broadly applicable, time- and cost-saving, synthetic routes to provide rapid access to compounds for subsequent use in drug discovery and development, thus contributing to the promotion of human health."
The project, titled "Catalytic Reductive C-H and C-C Silylation with Silyl Acetals," is funded by a three-year, $424,720 grant from the National Institute of General Medical Sciences, one of the National Institutes of Health.
Current methods to prepare these organosilanes utilize multi-step procedures which demand considerable time and effort. The project's goal is the design and application of single-pot, transition metal-catalyzed reductive C-H and C-C silylations for the synthesis of a new class of organosilanes.
A single-pot, or one-pot, synthesis improves the efficiency of a chemical reaction by subjecting a compound to successive chemical reactions using a single reactor. This method is preferred by chemists because it avoids a lengthy separation process and purification of the intermediate chemical compounds which saves time and resources while increasing chemical yield.
Jeon said the research will deliver several positive outcomes.
"There are three main aims of the research," he said. "In the first two, we will develop a new, simple, catalytic strategy for breaking an ubiquitous yet inert carbon-hydrogen bond and installing a new highly useful silicon moiety. This will allow for the rapid preparation of bio-medically important drugs.
"The third aim will provide a new tool to selectively break a strong carbon-carbon bond over adjacent, abundant carbon-hydrogen bonds and incorporate a silicon moiety. This method will be highly useful to prepare biologically active, secondary metabolites and pharmaceuticals."
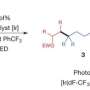
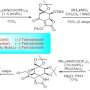
The project has already yielded one paper, titled "Catalytic Reductive ortho-C—H Silylation of Phenols with Traceless, Versatile Acetal Directing Groups and Synthetic Applications of Dioxasilines", which was published in the Journal of the American Chemical Society in June. Jeon co-authored the paper with Yuanda Hua, a UTA postdoctoral researcher; and two of the graduate students who work in his laboratory, Parham Asgari and Thirupataiah Avullala.
Jeon explained that during the past three decades, significant advances have been made to the organic processes of selectively transforming ubiquitous yet inert carbon-hydrogen bonds to other useful chemical entities. One reliable strategy for selective carbon-hydrogen bond cleavage within an organic is the use of a transition metal catalyst and a directing group, Jeon said. However, such a directing group is often difficult to install and manipulate after processes are completed.
"In this paper we describe how we first developed a general strategy for transition metal catalyst-assisted, site-selective carbon-hydrogen bond cleavage and insertion of a silicon moiety to phenols with a traceless directing group," Jeon said. "We believe that our new carbon-hydrogen bond functionalization approach to exploit readily available phenols and silanes more efficiently and cleanly will ultimately allow the practical production of diverse, biomedically important phenol derivatives and silicon-based materials."
A traceless directing group means that after a specific group directs, or guides, a catalyst to a specific position of a carbon-hydrogen bond within phenol, the group disappears without a trace. Phenols are a group of organic compounds whose aromatic ring is bonded to an alcohol group. Because phenol contains an alcohol group, it is able to form a very strong hydrogen bond with other molecules and often displays significant biological activities. Silanes are the basic building blocks of silicon chemistry and are versatile materials used in a wide range of applications.
Fred MacDonnell, professor and chair of the UTA Department of Chemistry & Biochemistry, said that Jeon's research has the potential to provide major positive impact on health and the human condition, one of four main themes in UTA's Strategic Plan 2020: Bold Solutions | Global Impact.
"Dr. Jeon's work is really starting to get a lot of attention from the scientific community and speaks to his creativity in this very challenging research area," MacDonnell said.
Jeon joined UTA in 2011. He earned a bachelor's of science degree in Chemistry in 2000 and a master's degree in Organic Chemistry in 2002, both from Sungkyunkwan University in Seoul, South Korea. He then came to the United States and did doctoral work at the University of Minnesota, receiving his Ph.D. in Organic Chemistry in 2009. After a two-year stint as a postdoctoral fellow at the University of Pennsylvania, he came to UTA.
More information: Yuanda Hua et al. Catalytic Reductive-C–H Silylation of Phenols with Traceless, Versatile Acetal Directing Groups and Synthetic Applications of Dioxasilines, Journal of the American Chemical Society (2016). DOI: 10.1021/jacs.6b04018
Journal information: Journal of the American Chemical Society
Provided by University of Texas at Arlington