Study finds straightforward way to model growth of vein networks
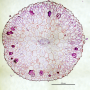
![(a) Immunostained mouse pup retina 7 days after birth. The hierarchical vascular network is clearly visible. Reprinted from [27], with permission. (b) Chemically cleared and stained leaf of Protium wanningianum. The first two levels of vascular hierarchy are marked in green. (c) Network representation of vascular flow. Edges (red, with lengths Le) carry flows Fe (arrows). At nodes (orange, representing areas ai), net currents Si (dark red arrows, only one shown for clarity) are drawn from the network, supplying the surrounding tissue. (d-f) Sketch of vascular development. Overlaid in grey is a co-moving coarse-graining grid. At the beginning (d), a tightly meshed capillary plexus is formed (capillaries in red). During development, the plexus is pruned and a hierarchical vascular tree is formed either due to (e) increasing perfusion, or (f) growth. Credit: arXiv:1606.00331v4 [physics.bio-ph] 22 Sep 2016 Study finds straightforward way to model growth of vein networks](https://scx1.b-cdn.net/csz/news/800a/2016/57f24162a3012.jpg)
Evolution tends to create simple rules that can work in organisms of all kinds. These rules are often all that nature needs to build some of the most complicated parts of biology through thousands of years of adaptation.
Vascular systems, for example, must efficiently deliver nutrients throughout an organism's body but must be adaptable to allow those organisms to grow into different shapes and sizes.
A new study by Eleni Katifori, an assistant professor in the Department of Physics & Astronomy of the University of Pennsylvania School of Arts & Sciences, and Henrik Ronellenfitsch, a postdoc in her lab, described how a simple rule guides the development of the vasculature in both animals and plants.
They used mathematical modeling to show that this rule, known as adaptive feedback, can produce the most efficient possible network of veins for any organism.
Their findings were published in Physical Review Letters.
The idea of adaptive feedback has been known in biophysics for some time. It refers to when plants and animals use the rate of liquid flow through their veins to decide to increase the size of the pathways and where to get rid of them.
"It's a very simple rule," Katifori said. "You use the vessel or you lose it."
A model using the rule of adaptive feedback alone, however, wasn't able to predict the kind of efficient vascular systems that appear in real-life organisms. There was a discrepancy in the field between development models predicted by adaptive feedback and the systems predicted to be most efficient.
Ronellenfitsch theorized that using adaptive feedback alone was not enough to predict how veins develop, and he set out to find what other variables he needed to explain it. It only took one other factor to set the model straight: growth. Ronellenfitsch found that, when he factored in the growth of an organism over time, the model fit real-world vein networks.
"Once I had the derivation, the rest was almost straightforward," Ronellenfitsch said. "It came as a surprise that it works as well as it does."
The model he devised not only reflected real life vein networks but also was the most efficient possible design. It predicts that, as an organism grows, a series of smaller and smaller veins spread out to carry nutrients to the new areas.
"You start off with a main vein that develops first," Ronellenfitsch said," and then secondary veins that branch off of the main vein development, third order veins that branch off of those, and so on and so on."
Katifori thinks this finding is especially remarkable as it can be generalized across animal and plant species. Because these rules are so ubiquitous in biology, she predicts that this model could have implications beyond the biological world.
It is likely that this model can be applied to other systems that distribute resources through a network, such as a power grid running electricity through its system.
"This study implies," Katifori said, "that whenever you have a system that transports liquids, energy, and maybe other things we haven't looked at, if you have growth then you will get something close to the best possible network—the absolute best one, not just a good one."
While this model helps explain how vein networks form, it does not provide the whole picture. The model does not yet include redundant veins that make it possible to recover from tissue damage, which is an important feature of biological systems.
"In our model, if you cut something, everything downstream from that vein is dead," said Ronellenfitsch, "and that's not a feature that vein networks have. Real networks are highly redundant."
Future research from the group will go toward understanding how different organisms build in redundancies, which could help further explain the underlying rules of evolution.
More information: Henrik Ronellenfitsch et al. Global Optimization, Local Adaptation, and the Role of Growth in Distribution Networks, Physical Review Letters (2016). DOI: 10.1103/PhysRevLett.117.138301 , arxiv.org/abs/1606.00331
Journal information: Physical Review Letters
Provided by University of Pennsylvania