Japanese research team elucidates structure of bacterial flagellar motor protein
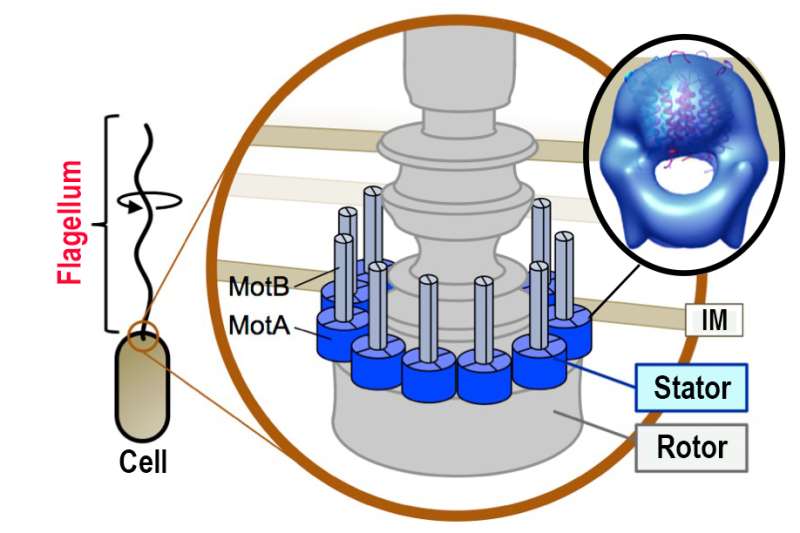
Many bacterial species use spiral propellers (flagella) attached to motors to move through a liquid environment. An interaction between the rotor and stator components of the motor generates the rotational force required for movement. The stator converts electrochemical energy into mechanical force after undergoing a structural change caused by a movement of charged particles (ions) through an internal channel. Previous studies investigated the stator and its interaction with the rotor by constructing mutant proteins and analyzing their functions. However, little was known about stator structure.
A team of Japanese researchers led by Homma's laboratory of Nagoya University have now purified the stator protein MotA from a bacterium found in hot springs (Aquifex aeolicus) and analyzed its three-dimensional structure using electron microscopy mainly in cooperation with Namba's laboratory of Osaka University. They found that it can form a structure of four MotA molecules (called a tetramer), which differs in shape from the previously predicted complex. The study was recently published in Scientific Reports.
The MotA protein spans the bacterial membrane, and has previously been shown to form a tetramer complex with another transmembrane protein, MotB, creating the stator. In this latest work, MotA was expressed and purified from A. aeolicus, and found to be structurally stable. Assessment of its interactive potential revealed it can form a tetramer even in the absence of MotB.
Electron microscopy showed that the elongated top part of the MotA complex matches the size of the lipid bilayer of the bacterium, suggesting that it represents the transmembrane component. "This region has a globular shape that corresponds to a MotA tetramer fitted inside an aggregate of detergent molecules which were used to purify the protein," first author Norihiro Takekawa says.
The lower part of the complex has two arch-like regions with spiky projections. "These match the cytoplasmic domain of the MotA protein," corresponding author Michio Homma says. "We predict that its structure will change with the movement of ions through the stator channel and in association with the stator-motor interaction." The shape of the complex differs from that reported for a related protein complex in another bacterium.
The article, "The tetrameric MotA complex as the core of the flagellar motor stator from hyperthermophilic bacterium" was published in Scientific Reports.
More information: Norihiro Takekawa et al, The tetrameric MotA complex as the core of the flagellar motor stator from hyperthermophilic bacterium, Scientific Reports (2016). DOI: 10.1038/srep31526
Journal information: Scientific Reports
Provided by Nagoya University



















