Removing cellular bookmarks smooths the path to stem cells
In reading, a bookmark tells where you stopped. Cells use bookmarks too, specific proteins that help the cell remember what collection of genes needs to be turned on again after the brief halt of gene expression during cell division. University of Alabama at Birmingham researchers are exploring the implications removing those bookmarks has on the promise of stem cells.
While the more than 200 different types of human cells all have the same genome, each cell type expresses a different ensemble of genes. These expressions of specific sets of genes make a neuron distinct from a muscle cell, a fibroblast cell—a cell in connective tissue that produces collagen and other fibers—or any other type of cell.
Now UAB researchers have found that removing these transcriptional bookmarks may be a key to better reprogramming of human fibroblasts to create induced pluripotent stem cells, or iPS cells. An iPS cell is generated by epigenetic modulation from any somatic cell—usually skin or blood cells from a child or adult—to have it behave like an embryonic stem cell. As the name implies, these cells are pluripotent, which means they have the ability to form all adult cell types.
Kejin Hu, Ph.D., an assistant professor in the UAB Department of Biochemistry and Molecular Genetics, calls this de-bookmarking or de-reading. He is able to de-bookmark using small-molecule chemicals that mildly target the binding pockets of the bookmarking proteins, known as bromodomains extra terminal, or BET. The result, Hu says in a paper published in Cell Reports on Sept. 20, is a proof-of-principle strategy to facilitate reprogramming to pluripotency.
Improved reprogramming offers two benefits. First, it may increase the yield of iPS cells created from human fibroblasts, a yield that currently is much lower than reprogramming of mouse cells. Second, it may improve the quality of the iPS cells by ensuring that more of the somatic genes—those expressed in a differentiated cell, such as a fibroblast—are efficiently turned down or turned off during reprogramming to the iPS cells.
Quality reprogramming is a vital step in efforts to use iPS cells in medical research and disease treatments. Human iPS cells—which have the ability to differentiate into any type of specialized cell—may be able to transform transplantation medicine by creating patient-specific cell-replacement therapies to treat neurological diseases, heart ailments, blood diseases and diabetes.
"Human cells have more than 40,000 genes, but only a portion of the genome is expressed in a specific cell type," Hu said. "This expression of a specific set of genes defines the cell identity. To establish a new cell type—in my case, a pluripotent stem cell—we have to erase the old program of gene expression, in addition to establishing the new transcriptional program specific for iPS cells. It's like construction: If you are going to build a new building on the same site, you have to remove the old one first."
The reprogramming factors that are commonly used to create iPS cells from fibroblasts—based on the breakthrough work of Nobel laureate Shinya Yamanaka in 2006—face a reprogramming barrier, Hu says.
"If we can lower the barrier, we can enhance the reprogramming efficiency," Hu said. "My strategy is to use chemicals to erase the transcriptional program specific to the starting cells."
Hu found that a very low concentration of JQ1, an inhibitor of the BET family of proteins:
- Down-regulated 390 fibroblast-specific genes when applied to naïve human fibroblasts
- Down-regulated 651 fibroblast-specific genes when applied to human fibroblasts during reprogramming
- Increased the efficiency of successful reprogramming of human fibroblasts to iPS cells by 20-fold
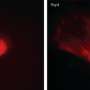
In support of this gene expression and reprogramming data, fibroblasts change shape when they are treated with JQ1, from a long spindle shape to polygonal or rounded cell, which shows loss of fibroblast identity and transition to pluripotent stem cells. Presumably, genes that are needed to maintain the spindle shape are down regulated by JQ1.
Hu proposes the following model to explain his results. During normal cell division, the active fibroblast genes are bookmarked by attachment of BET proteins to acetylated chromatin during the mitotic phases, while RNA Polymerase II drops off of the chromatin. At the start of interphase, these bookmarks guide the polymerase back to the genes, and they again are transcribed by RNA Polymerase II. In contrast, when JQ1 is added at low concentration, the active fibroblast genes are de-bookmarked by the interaction of JQ1 with the BET proteins during the mitotic phases of cell division. This 'erases' epigenetic memory of the fibroblast gene expression, which in turn results in loss of fibroblast gene transcription when interphase returns. This also increases the success of reprogramming into pluripotent stem cells.
More information: "Reprogramming by de-bookmarking the somatic transcriptional program through targeting of BET bromodomains," Cell Reports, 2016. www.cell.com/cell-reports/full … 2211-1247(16)31141-X , DOI: 10.1016/j.celrep.2016.08.060
Journal information: Cell Reports
Provided by University of Alabama at Birmingham