Study yields insight into the mystery of smell

Researchers at the University of Pittsburgh School of Medicine have uncovered the mechanism underlying a phenomenon in how we smell that has puzzled researchers for decades. In an article appearing online today in the Proceedings of the National Academy of Sciences, the team reports that, surprisingly, the mechanism follows a simple physics principle called cooperativity.
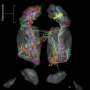
Inhalation of a scent sends a complex mixture of odor molecules swirling toward the back of the nose, where they bind to specialized receptors that are located on millions of olfactory neurons. Activation of these receptors sends signals from the olfactory neurons to the brain, where the smell is deciphered.
Individual neurons have only a single type of receptor and, therefore, recognize only specific odor molecules. However, the hundreds of different types of olfactory receptors are found, or expressed, in approximately equal numbers across the entire population of neurons, which allows a person to detect a wide variety of smells, explained senior investigator Jianhua Xing, Ph.D., associate professor of computational and systems biology, Pitt School of Medicine. Richard Axel, Columbia University, and Linda Buck, now at the Fred Hutchinson Cancer Research Center, received the 2004 Nobel Prize in Physiology or Medicine for discovering the receptors and making these observations.
"Over the past decades, neuroscientists have been trying to uncover how nature accomplishes these two goals: selecting one, and only one, type of olfactory receptor for each neuron, while at the same time ensuring that all receptor types are represented in the whole population of neurons," said Dr. Xing.
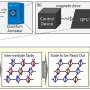
The mysteries of how we smell have generated many experimental observations about how olfactory receptors actually work. In the new study, Dr. Xing and colleagues used these existing experimental data to create a computational model of how olfactory receptor expression can be both uniform across a single neuron, yet very diverse across the entire population of neurons. They then used this model to correctly predict several additional findings that have been demonstrated by other research groups, demonstrating that their model is valid.
Surprisingly, the model suggested a three-pronged regulation of olfactory receptor gene expression that follows a basic physics principle called cooperativity, in which elements in a system influence the behavior of one another rather than function independently. Cooperativity can explain many phenomena, such as the transition between liquid and vapor states, why oil and water do not mix, and even other biological processes such as how a protein folds.
"We are amazed that nature has solved the seemingly daunting engineering process of olfactory receptor expression in such a simple way," said Dr. Xing.
The findings pave the way for new predictions about how olfactory receptors function that can be tested in future experiments, the results of which will help the team refine their model and make even more predictions.
More information: Achieving diverse and monoallelic olfactory receptor selection through dual-objective optimization design, PNAS, www.pnas.org/cgi/doi/10.1073/pnas.1601722113
Journal information: Proceedings of the National Academy of Sciences