Scientists track charge in future fuel cell material
Scientists have mapped the movement of charge through a designer material for the first time by using a combination of X-ray and neutron techniques. The material, known as a metal organic framework or "MOF", has been designed to act as a host of charge in future fuel cells, which offer a greener alternative to combustion engines in vehicles. Now that scientists can better understand the mechanism behind the flow of charge in the MOF, they can turn to designing more efficient fuel cells in the pursuit of cleaner energy.
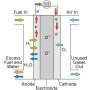
Fuel cells convert the chemical energy of a fuel into electricity by means of a chemical reaction. In transportation there are a variety of fuel cells available, with polymer electrolyte membrane (PEM) fuel cells currently used most widely.
However, the performance of a fuel cell strongly depends on the efficiency of the electrolyte, the porous material at the centre of the fuel cell that controls the flow of charge between the positive and negative electrodes. To boost fuel cell efficiency, scientists are developing smart electrolyte membranes with new designer materials that produce the most charge.
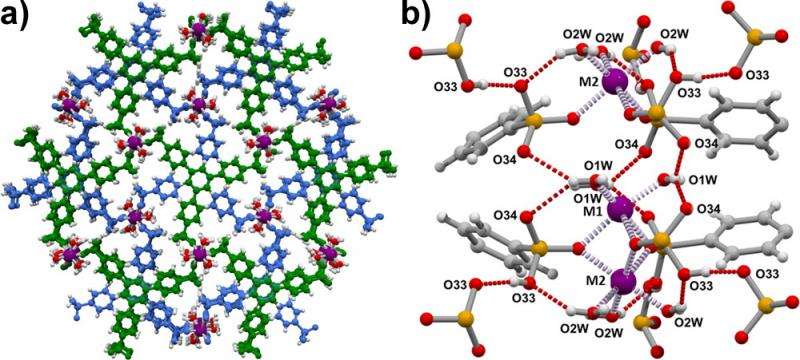
These scientists include a team from Manchester and Nottingham Universities who are designing molecular networks known as MOFs to act as the source of charge in the membrane of a fuel cell. Using X-rays at Diamond Light Source, the UK's synchrotron science facility, the scientists were able to map the atomic structure and workings of the MOF. The team then used neutrons at the UK's Science and Technology Facilities Council's (STFC) ISIS Neutron and Muon Source to map the flow of charge, known as proton conduction, in a MOF for the first time. They found the protons move freely within a sphere, rather than the more common "jump diffusion between sites" mechanism. The results have been published in the Journal of the American Chemical Society. This new knowledge will enable the design of improved proton conducting materials by optimising the pathway the protons take.
Drs Sihai Yang and Martin Schröder, group leaders from the University of Manchester, say,
"The success of this work will not only create new knowledge on MOFs, but also enable the building of next generation fuel cells that operate at a wider temperature range and with higher efficiency. This will help bring this technology into a reality."
MOFs are a new family of hybrid materials made up of metals connected by organic linkers called ligands. By substituting different metals and ligands in the MOF, scientists can control its function. In this case, the MOF, known as MFM-500(Ni) is made up of nickel and phosphonate, a salt of phosphoric acid that produces the protons.
Dr Simona Pili, lead author from the University of Manchester, designs and makes the MOFs in the laboratory. She says,
"The great thing with MOFs is that you can design the ligand to have different functional groups. We want to use ligands with an excess of protons already present, to provide more protons for the free movement within the MOF. Once we know what we need, we can go back to the lab and improve on the design."
Scientists designing new materials that are going to function effectively in real life processing situations need to test them using a range of experimental techniques. The team studying these MOFs used X-rays and neutrons, two complementary techniques to study the material at the atomic level. The team visited STFC's ISIS Neutron and Muon Source, a "super-microscope" and used neutrons to map the movement of charge within the MOF. ISIS scientist and co-author of the study,
Dr Ian Silverwood says, "At ISIS, a technique known as quasi-elastic neutron scattering allows us to look at how atoms move on a timescale of picoseconds, or trillionths of a second. We use it to follow the mechanism of proton conduction, watch diffusion in solids and liquids and see how molecules move and re-orientate themselves in complex systems such as polymers and proteins. The IRIS instrument is a spectrometer that uses arrays of highly ordered crystals to analyse how neutrons have changed energy and momentum after interacting with a sample, such as the MOF in this study. This gives us a picture of the direction the atoms are moving in, and by how much. The uniquely strong interaction of neutrons with hydrogen makes them the ideal probe for this system."
The team also used the intense X-rays generated at Diamond Light Source, the UK's synchrotron science facility. Both the Powder Diffraction (I11) and Small Molecule Diffraction (I19) beamlines were used to explore the atomic make-up of the new MOF material and determine how it behaves at an atomic level when exposed to real life conditions such as high temperatures.
Dr Claire Murray, Support Scientist on Diamond's Powder Diffraction beamline explains, "This research represents really beautiful chemistry in a new and exciting area of MOF research. Up until now, MOFs have been mainly used for gas absorption but scientists are hoping to be able to exploit proton conductivity for the production of green energy fuel cells that can operate at high temperatures and in dry conditions. Because the X-rays we generate at Diamond are so intense, researchers can reveal the structure and workings of materials such as MFM-500(Ni) in very fine detail, enabling them to detect tiny changes that could impact on the performance of the material. This complemented the team's neutron experiments at ISIS where they were able to study the proton movement in the framework. This is very exciting as it has never been done before. We are now looking forward to further experiments at Diamond as the team advance their studies by putting the MOFs into the membranes of fuel cells and testing them under conditions that mimic those they would be operating under in real life."
Now that the team have cracked the mechanism behind proton conductivity in the MOF, it's back to the laboratory to use this information to design and study new materials. Dr Pili and the group are focusing on trying to increase the conductivity of the material, either by improving the ligands or doping the MOF with gases that are rich with protons.
More information: Simona Pili et al. Proton Conduction in a Phosphonate-Based Metal–Organic Framework Mediated by Intrinsic "Free Diffusion inside a Sphere", Journal of the American Chemical Society (2016). DOI: 10.1021/jacs.6b02194
Journal information: Journal of the American Chemical Society
Provided by Science and Technology Facilities Council