New synthetic route to potential medicines for type 2 diabetes
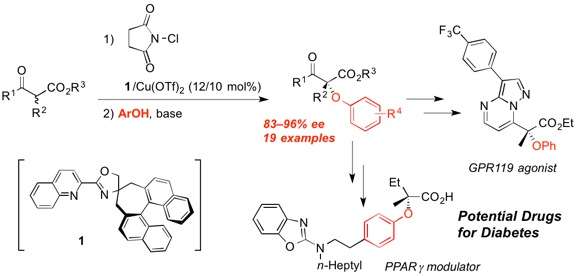
Aryl alkyl ethers are important structural motifs found in many biologically active compounds. Therefore, stereoselective etherification is a highly important synthetic operation in the preparation of drug candidates. However, very few enantioselective methods have been described for the synthesis of chiral tertiary aryl ethers.
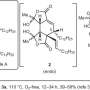
Researchers at Toyohashi Tech have found that the SN2 reaction of α-chloro-β-keto esters with phenols proceeded smoothly despite the fact that the reaction occurred at a tertiary carbon. They previously reported the highly enantioselective chlorination of β-keto esters with a chiral Lewis acid catalyst. Thus, in the two investigations, they have successfully demonstrated the enantioselective phenoxylation of β-keto esters. The novel presented method allows the synthesis of α-aryloxy-β-keto esters with high enantioselectivity.
"Etherification by the SN2 reaction is an older synthetic method called Williamson ether synthesis, but very few researchers have succeeded in conducting this reaction with tertiary halides," said Associate Professor Kazutaka Shibatomi. "This is the first example of the enantioselective synthesis of α-aryloxy-β-keto esters, which would be useful synthetic intermediates for new drug candidates."
Using this method, the researchers demonstrated the synthesis of some biologically active compounds, such as a GPR119 agonist and a PPARγ modulator, for the potential treatment of type 2 diabetes. The researchers expect that the present method will also be helpful in preparing other types of synthetic drugs.
More information: Kazutaka Shibatomi, Manato Kotozaki, Nozomi Sasaki, Ikuhide Fujisawa, and Seiji Iwasa (2015). Williamson ether synthesis with phenols at a tertiary stereogenic carbon: formal enantioselective phenoxylation of β-keto esters, Chemistry - A European Journal, Article published online before print, 10.1002/chem.201502042
Kazutaka Shibatomi, Yoshinori Soga, Akira Narayama, Ikuhide Fujisawa, and Seiji Iwasa (2012). Highly enantioselective chlorination of β-keto esters and subsequent SN2 displacement of tertiary chlorides: a flexible method for the construction of quaternary stereogenic centers, Journal of the American Chemical Society, 134(24), 9836-9839, 10.1021/ja304806j
Journal information: Chemistry – A European Journal , Journal of the American Chemical Society
Provided by Toyohashi University of Technology