New temperature record: Hydrogen sulfide becomes superconductive under high pressure at minus 70 degrees Celsius

Up until now, no material has been able to conduct current with no resistance at such high temperatures: Researchers at the Max Planck Institute for Chemistry in Mainz and the Johannes Gutenberg University Mainz observed that hydrogen sulfide becomes superconductive at minus 70 degree Celsius—when the substance is placed under a pressure of 1.5 million bar. This corresponds to half of the pressure of the earth's core. With their high pressure experiments the researchers in Mainz have thus not only set a new record for superconductivity—their findings have also highlighted a potential new way to transport current at room temperature with no loss.
For many solid-state physicists, superconductors that are suitable for use at room temperature are still a dream. Up until now, the only materials known to conduct current with no electrical resistance and thus no loss did so only at very low temperatures. Accordingly, special copper ceramics (cuprates) took the leading positions in terms of transition temperature—the temperature at which the material loses its resistance. The record for a ceramic of this type is roughly minus 140 degrees Celsius at normal air pressure and minus 109 degrees Celsius at high pressure. In the ceramics, a special, unconventional form of superconductivity occurs. For conventional superconductivity, temperatures of at least minus 234 degrees Celsius have so far been necessary.
A team led by Mikhael Eremets, head of a working group at the Max Planck Institute for Chemistry, working in collaboration with the researchers at Johannes Gutenberg University Mainz has now observed conventional superconductivity at minus 70 degrees Celsius, in hydrogen sulfide (H2S). To convert the substance, which is a gas under normal conditions, into a superconductor the scientists did however have to subject it to a pressure of 1.5 megabar (1.5 million bar), as they describe in the latest edition of the science magazine Nature.
The transition temperature of conventional superconductivity knows no limits
"With our experiments we have set a new record for the temperature at which a material becomes superconductive," says Mikhael Eremets. His team has also been the first to prove in an experiment that there are conventional superconductors with a high transition temperature. Theoretical calculations had already predicted this for certain substances including H2S. "There is a lot of potential in looking for other materials in which conventional superconductivity occurs at high temperatures," says the physicist. "There is theoretically no limit for the transition temperature of conventional superconductors, and our experiments give reason to hope that superconductivity can even occur at room temperature."
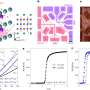
The researchers generated the extremely high pressure required to make H2S superconductive at comparatively moderate negative temperatures in a special pressure chamber smaller than one cubic centimeter in size. The two diamond tips on the side, which act as anvils, are able to constantly increase the pressure that the sample is subjected to. The cell is equipped with contacts to measure the electrical resistance of the sample. In another high-pressure cell, the researchers were able to investigate the magnetic properties of a material that also change at the transition temperature.
After the researchers had filled the pressure chamber with liquid hydrogen sulfide, they increased the pressure acting on the sample gradually up to roughly two megabar and changing the temperature for each pressure level. They took measurements of both resistance and magnetization to determine the material's transition temperature. The magnetization measurements provide very useful information, because a superconductor possesses ideal magnetic properties.
Hydrogen atoms facilitate superconductivity at high temperatures
The researchers believe that it is mainly hydrogen atoms that are responsible for hydrogen sulfide losing its electrical resistance under high pressure at relatively high temperatures: Hydrogen atoms oscillate in the lattice with the highest frequency of all elements, because hydrogen is the lightest. As the oscillations of the lattice determine the conventional superconductivity—and do this more effectively the faster the atoms oscillate—materials with high hydrogen content exhibit a relatively high transition temperature. In addition, strong bonds between the atoms increase the temperature at which a material becomes superconductive. These conditions are met in H3S, and it is precisely this compound that develops from H2S at high pressure.
Mikhael Eremets and his team are now looking for materials with even higher transition temperatures. Increasing the pressure acting on the hydrogen sulfide above 1.5 megabar is not helpful in this case. This has not only been calculated by theoretical physicists, but now also confirmed in experiments performed by the team in Mainz. At even higher temperatures the electron structure changes in such a way that the transition temperature slowly begins to drop again.
Wanted: hydrogen-rich materials with a higher transition temperature
"An obvious candidate for a high transition temperature is pure hydrogen," says Mikhael Eremets. "It is expected that it would become superconductive at room temperature under high pressure." His team has already begun experimenting with pure hydrogen, but the experiments are very difficult as pressures of three to four megabar are required.
"Our research into hydrogen sulfide has however shown that many hydrogen-rich materials can have a high transition temperature," says Eremets. It may even be possible to realize a high-temperature superconductor worth the name in terms of common temperature perception without high pressure. The researchers in Mainz currently need the high pressure to convert materials that act electrically insulating like hydrogen sulfide into metals. "There may be polymers or other hydrogen-rich compounds that can be converted to metals in some other way and become superconductive at room temperature," says the physicist. If such materials can be found, we would finally have them: superconductors that can be used for a wide range of technical applications.
More information: "Conventional superconductivity at 203 K at high pressures." Nature, August 17, 2015. DOI: 10.1038/nature14964 . Available at: arxiv.org/abs/1506.08190
Journal information: Nature
Provided by Max Planck Society