Scientists develop better way to visualize molecules
Computer scientists at the University of Toronto Scarborough campus have solved a core problem in strucutral biology by developing a faster, cheaper and more reliable way to determine the three-dimensional shapes of biological molecules such as proteins and viruses.
Simplifying the modeling process makes it vastly more possible to understand these molecules' function and behaviour, which is important for both basic science and cutting-edge development of new drugs and medicines, according to UTSC postdoctoral fellow Marcus Brubaker (pictured above).
"At a high level, these molecules look kind of like everyday shapes," he says. "At a fine level, though, they're very detailed. It's like trying to recreate the 3-D structure of a boxful of smoke."
Brubaker and two other UTSC computer scientists recently presented their new method at the 2015 IEEE Conference on Computer Vision and Pattern Recognition in Boston.
The physical shape of biological molecules largely determines their behaviour. They interact like tiny locks and keys, meaning a protein can only react to an enzyme, drug or other biomolecule if the two have compatible contours.
Studying these microscopic structures can be painstaking: biological molecules are as delicate as they are intricate, and many potentially useful scanning methods risk damage or destruction before any useful information can be gleaned.
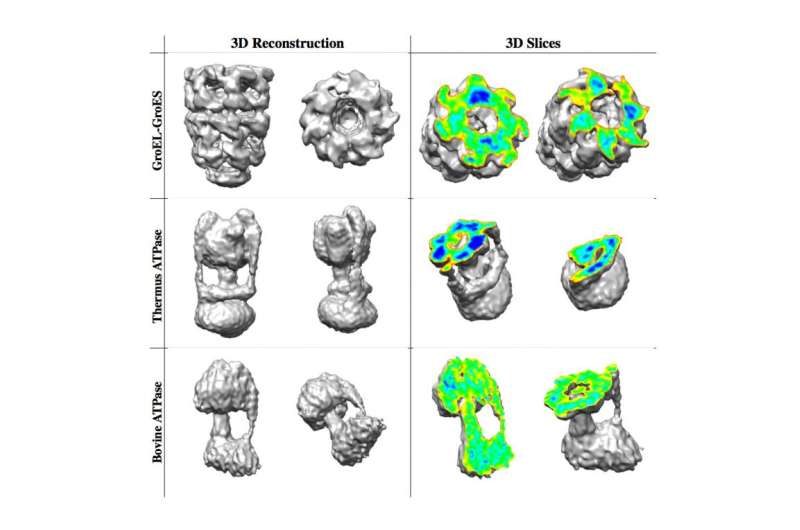
The researchers created thousands of two-dimensional images of a molecule, using a technique called cryo-electron microscopy. The difficulty lay in reconstructing a three-dimensional molecular architecture from these flat images.

"There are many challenges in estimating the 3D structure, but the big one for us is that we don't know from which direction those 2D images were taken," says Brubaker. "We don't know how they are oriented with respect to each other."
The first steps of their iterative method involve a time-consuming evaluation for every possible orientation for each 2D image. But as the structure takes shape, more and more possibilities can be rejected as statistically unlikely. The process accelerates with each iteration, allowing a single computer to achieve in a day what previously would have required an array of hundreds of computers and several weeks.
Brubaker and his colleagues are considering creating a spin-off company, but they see the value of this advance as being greater than any commercial return.
"We were initially interested because it's a challenging computational problem," Brubaker says. "But we've been fortunate to collaborate with a biologist at Toronto's Hospital for Sick Children who helped us understand the way this could be used. We're thinking about how to make this discovery useful to the community."
Provided by University of Toronto