May 1, 2015 report
Metal-organic framework with a fixed composition despite initial concentration of components
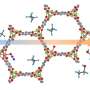
![MOF-2000 is a periodic framework harboring controlled component heterogeneity. Chemical structures of organic struts H2Lr and H2Lb4+ incorporated in MOF-2000 (Methods Summary), and the MOF-2000 crystal structure [key: carbon, black; oxygen, red; Zn4O(CO2)6 polyhedra, blue; all hydrogen atoms have been omitted for clarity]. Credit: (c) 2015 PNAS, 10.1073/pnas.1416417112 Metal-organic framework with a fixed composition despite initial concentration of components](https://scx1.b-cdn.net/csz/news/800a/2015/55436ae3bc752.png)
(Phys.org)—Metal-organic framework (MOF) chemistry is a multi-disciplinary field that connects organic and inorganic chemistry with materials science and nanotechnology. A key feature of MOFs is the self-assembly of organic molecules around metal ions or metal clusters. The composition of the MOFs is often dependent on the initial conditions in which the components are combined.
The goals of MOF design are to make a compound whose characteristics, such as pore size, can be tuned using a variety of building blocks, has well-defined, predictable connectivities, and is robust enough to form under different environmental conditions. In other words, the goal is to obtain the versatility of biological compounds with the regularity of covalent molecules. Andrew C.-H. Sue, Ranjan V. Mannige, along with a team from the Departments of Chemistry at The University of California in Berkeley, the Molecular Foundry at Lawrence Berkeley National Laboratory, and Northwestern University report a new MOF that is environmentally robust, maintaining a 2:1 ratio of components, despite their initial solution concentration. Their work is reported in Proceedings of the National Academy of Sciences.
Sue, Mannige, et al. have designated their new framework as MOF-2000. It is comprised of organic struts with the same backbone and but with side chains that are either a crown ether or a catenane. Both crown ether and catenane are well-known classes of structural motifs, and the backbone described has aromatic components. The struts are coordinated with Zn4O(CO2)6. The structure is isotropic, or the same in all directions, and nonperiodic.
Experimental results showed that the MOF-2000 displays a 2:1 ratio of crown ether-to-catenane, even when the ratios of these components in the parent solution are varied by an order of magnitude. This pattern is unprecedented in MOF chemistry. Usually the organic struts combine in ratios based on initial concentrations in the parent solution, but this one does not. In this study, the authors investigated why the MOF-2000 maintained this "magic number" ratio in hopes of finding general characteristics that can be applied to other system designs.
The authors subjected MOF-2000 to theoretical modeling, Monte Carlo simulations, and crystal studies to determine the mechanism behind this ratio. Modeling indicated that the 2:1 ratio is not due to thermodynamic effects, as the thermodynamic product would likely have components in a 1:1 ratio. Monte Carlo simulations indicate that this ratio appears in energetically minimal pockets in a non-equilibrium state. Additionally, the consistent ratio does not seem to be due to charge complementarity or from thermodynamically favorable close-packing.
Simulations indicate that this framework is a kinetic product, and the lower energy state may be due to noncovalent interactions. Crystal studies in which 1:1 ordered domains and grain boundaries were investigated show that this ratio may also be a function of the framework topology. The thermodynamic product is a 1:1, antiferromagnetic structure and growth simulations indicate that a 2:1 structure arises when the thermodynamically stable 1:1 catenane/crown ether pattern is interrupted by crown ether-crown ether "mistakes." This 2:1 structure preference may arise from kinetic trapping that occurs during the attempted formation of the thermodynamic product, and suggests that there is a hierarchy of noncovalent interactions.
Overall, these intriguing findings indicate that this magic number ratio, which produced a concentration-independent MOF, occurs in systems where certain noncovalent interactions are energetically favorable compared to other noncovalent interactions in an assymetric fashion. This was seen in the favorable crown ether-catenane interaction in the MOF-2000 system. Also, the system must allow for reversible interactions of assembly "mistakes" during the crystallization process.
More information: "Heterogeneity of functional groups in a metal-organic framework displays magic number ratios" PNAS, 10.1073/pnas.1416417112
Abstract
Multiple organic functionalities can now be apportioned into nanoscale domains within a metal-coordinated framework, posing the following question: how do we control the resulting combination of "heterogeneity and order"? Here, we report the creation of a metal–organic framework, MOF-2000, whose two component types are incorporated in a 2:1 ratio, even when the ratio of component types in the starting solution is varied by an order of magnitude. Statistical mechanical modeling suggests that this robust 2:1 ratio has a nonequilibrium origin, resulting from kinetic trapping of component types during framework growth. Our simulations show how other "magic number" ratios of components can be obtained by modulating the topology of a framework and the noncovalent interactions between component types, a finding that may aid the rational design of functional multicomponent materials.
Journal information: Proceedings of the National Academy of Sciences
© 2015 Phys.org