Biofuels and chemicals that don't cut into the food supply
Emma Master's team spent the last two years studying plant cell walls, the part of the cell that gives trees and other flora their structural strength. The wall itself is built from a tight complex of sugars and polymers, and Master wants to isolate these components so they can be used to create renewable fuels, high-value chemicals, and novel bio-based materials.
Sugar is much more than just a sweetener, and the cell wall's sugar could be a source for making valuable renewable bioproducts, including fuel. It's an intriguing possibility, using inedible plant parts to make cleaner energy.
Master heads up the University of Toronto Bioproducts Research Lab which focuses on developing enzymes to create new polymers and chemicals from plant fibre. The cell wall is mainly made up of three different polymers, or molecule chains, that each contribute to its overall strength. A major component is cellulose, a chain of glucose sugars, which is especially sought-after for making biochemicals and biofuels.
In particular, biofuels make headlines regularly as a potential green alternative to coal and gas energy—a chance to use replenishable crops to meet our energy needs rather than non-renewables from oil sands and fracking.
Now, some biofuels are already on the market, but most options today are made from starches which humans can eat. Proponents would love to see a reliable and economical source for glucose that doesn't cut into the food supply.
That's where Master's research comes in. Her team looks at how to release the cell wall polymers, so they can be usefully turned into renewable materials and chemicals, as well as fuel.
"If you're thinking about liquid biofuels for transport in your car for instance, the challenge is how to economically get that cellulose out of the cell wall and then how to convert this polymer into single sugars," said Master.
Once released from the cell wall, single sugars can be converted to biofuel by fermenting microorganisms like yeast and bacteria, which in turn can be used as an alternative fuel source in cars and industry.
Decades of research have identified key enzymes that break down plant matter into fermentable sugars, but it remains a costly procedure, in part because of how difficult it is to reach the sugar chains encased in other polymers in the cell wall.
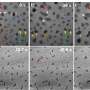
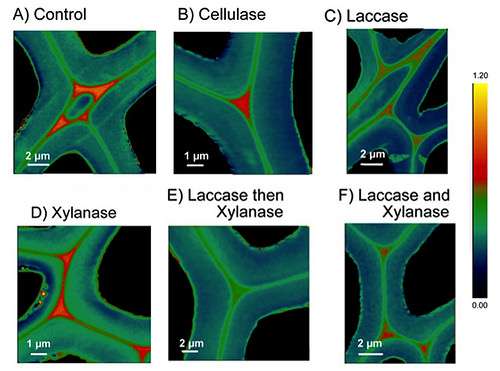
To overcome this hurdle, Master's team focused on analyzing how different enzymes worked together to break down the cell wall, to understand what worked best, and why. For a long time, they looked at cell wall chemistry using of Time-of-flight Secondary Ion Mass Spectrometry (ToF-SIMS), but were interested in achieving a higher level of spatial detail.
A post-doctoral fellow on the project had worked at the Canadian Light Source before, and recommended the facility for its ability to give extremely detailed pictures of chemical changes taking place in a sample. The change would effectively be like changing from regular TV to high definition.
Over three visits to the CLS, the team carried out studies to understand how various enzymes work to break down the cell wall, both individually and as protein cocktails.
The process was a steep learning curve, as the team had to learn brand-new ways to prepare their samples and even interpret their data.
"I have to say what kept us going was that the group I was working with were as passionate as me about the science and understood the patience needed to address big questions," said Master.
Those questions went beyond even working towards an ideal enzyme cocktail for making sustainable biofuels. Master sees the work as a way to unlock genetic code.
Every enzyme is a protein produced by some organisms's genetic code. As Master and others decode the functions of specific enzymes, Master hopes that genomic researchers will be able to better understand how certain genes evolved.
This latest publication is only a part of the story, and the Master research group plans to come back to the CLS to continue their cell wall work. Their next project involves looking at enzymes that apply a unique mechanism to breaking through cellulose, which could reduce overall costs associated with plant fibre processing.
More information: Jeremic, Dragica, et al. "Direct and up-close views of plant cell walls show a leading role for lignin-modifying enzymes on ensuing xylanases." Biotechnology for Biofuels 7.1 (2014): 496. DOI: 10.1186/s13068-014-0176-9
Journal information: Biotechnology for Biofuels
Provided by Canadian Light Source