'Rewriting' the way to make natural drug compounds
One of the big hurdles in bringing drugs to market is the difficulty of producing large enough quantities of potential compounds to conduct clinical trials. This is particularly true with compounds made by organisms, which usually are produced in exceedingly small amounts and are difficult to synthesize in the laboratory.
Researchers from the University of Utah College of Pharmacy have made an important stride in addressing those problems. In a study in ACS Synthetic Biology, researchers led by Eric W. Schmidt, Ph.D., professor of medicinal chemistry, describe an inventive technique that allowed them to take the genes that instruct a species of fungi to make large quantities of a native compound, and then "rewrite" them to make a different compound of potential therapeutic importance.
The study shows that one way to solve problems of synthesis of natural compounds is to figure out how an organism solves the problem itself, and then modify it for a particular use, according to Schmidt, who also holds the William R. Droschkey Endowed Chair in Pharmacy.
"In terms of drug discovery, there remains a large gap between finding a compound that's a potential drug and bringing it to market," he says, "so we've been looking at synthetic biology—how to write and rewrite genetic code to produce these compounds for us."
The compound the researchers synthesized was discovered at Wyeth Pharmaceuticals, now part of Pfizer Inc., in collaboration with another U College of Pharmacy faculty member, Louis R. Barrows, Ph.D., professor of pharmacology and toxicology. The compound was discovered because of its anti-tuberculosis properties. Barrows now is investigating the compound for potentially developing a new TB drug. (This work was highlighted in the Journal of Natural Products.)
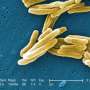
To make the compound, the researchers used a fungus, Fusarium heterosporum, that naturally produces a chemical, called equisetin, in large quantities. Schmidt and the study's first author, Thomas B. Kakule, a University of Utah doctoral student in Medicinal Chemistry, deleted the equisetin-making pathway in the fungus. Then they gave two genes essential for synthesizing the chemical new instructions to make the anti-TB compound. Whereas it was previously a struggle to harvest enough of the anti-TB compound to even determine its chemical make-up, using the new system they were able to make enough of the compound to completely characterize the chemical and its anti-TB properties.
Another desirable feature, which was already built into the natural instructions for making equisetin, is that the fungal factory is highly controllable. The pathway is normally "off", but is turned "on" when the fungi is grown on corn grits, which can be bought at a natural food store. This inducible system means that they can make large amounts of nearly any compound whenever they choose, even those that are normally toxic to the organism.
"Basically, we exploited the native machinery that makes compounds in the fungus and redirected it to make the compound we wanted," Schmidt says. "I don't know why it hasn't been done before. We just felt that tapping into a native pathway that's in tune with the metabolism of the cell would have its advantages."
Although the research is in its early stages, the results with fungus represent an important step in merging genetic engineering with chemistry to answer basic questions about how natural compounds are made. The model for turning fungus into a factory for making an anti-TB compound can be applied to other organisms to address different diseases and issues.
Journal information: ACS Synthetic Biology , Journal of Natural Products
Provided by University of Utah Health Sciences