Chemists develop porous molecules that bind greenhouse gases

A team of University of Houston (UH) chemistry researchers have developed a molecule that assembles spontaneously into a lightweight structure with microscopic pores capable of binding large quantities of several potent greenhouse gases.
"Greenhouse gases, such a carbon dioxide, have received much attention lately because of their potential to dramatically affect Earth's climate, primarily the temperature of the planet," said Ognjen Miljanić, a UH associate professor of chemistry and leader of the team.
While carbon dioxide presents the biggest problem, Miljanić notes that several other compounds are hundreds or thousands of times more potent in their greenhouse effect per unit of mass. These compounds include Freons, used as common refrigerants, and fluorocarbons, highly stable organic compounds in which one or more hydrogen atoms have been replaced with fluorine.
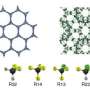
"We developed a molecule that self-assembles into a structure that can capture these greenhouse vapors to the tune of 75 percent by weight," Miljanić said. "This molecule could be used to capture Freons from disposed refrigeration systems, for example, or to concentrate them prior to analysis of their content."
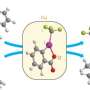
In their recent paper in Nature Communications, Miljanić and his colleagues report that a small molecule based on an extensively fluorinated backbone will form a structure with extremely small pores about 1.6 nanometers in diameter. Members of the team included Miljanić and professors Allan Jacobson and Olafs Daugulis, all from UH's Department of Chemistry in the College of Natural Sciences and Mathematics.
"These tiny pores are lined with fluorine atoms, giving them a high affinity for other molecules containing fluorine - such as fluorocarbons and Freons," Miljanić said.
Porous materials with similar pore sizes have been developed in previous studies, but those materials were often heavy, because of the presence of metals, as well as sensitive to water and difficult to process and recycle.
"The advantages of the current material is that it is stable to water and composed from individual molecules held together only by weak interactions," Miljanić said. "This latter feature makes this material lightweight, because there are no metal connectors."
The weak interactions between the molecules can be broken when needed, so the molecule can be recycled or deposited on a surface. The molecule is stable to 280 degrees Celsius.
In this international collaboration, UH researchers worked with Yu-Sheng Chen from the University of Chicago and Yu-Chun Chuang from the Taiwan National Synchrotron Radiation Research Center. A provisional patent based on this work has been filed.
Journal information: Nature Communications
Provided by University of Houston