New method: More pharmaceuticals could benefit from the stabilizing influence of fluorine atoms
Molecules with potent bioactivity alone are not suitable for new medicines. They must behave well inside the body, being easily absorbed, yet metabolically stable, once circulating through the blood stream. Adding fluorine atoms is a proven way to improve these properties. Now, this approach is set to expand thanks to a technique developed by Mikiko Sodeoka and her colleagues at the RIKEN Advanced Science Institute in Wako.
Sodeoka and her team's approach is to attach fluorine in the form of the trifluoromethyl group, -CF3, a small side chain that adds three fluorine atoms in a single transformation. Since trifluoromethyl groups are hydrophobic, they help the drug to infiltrate the body and reach its site of action. The carbon–fluorine bond is also very strong, improving the drug's stability. "These factors are crucial for the development of therapeutic drugs and agrochemicals," Sodeoka says.
Despite the advantages offered by trifluoromethyl groups, the narrow range of substrates to which they can be attached currently has limited their use. While methods for attaching the group at a carbon–carbon double bond are well established, only a few approaches exist for attaching the group at other sites on the molecule.
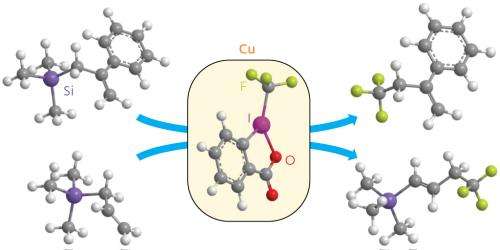
Sodeoka's strategy was to first functionalize the target molecule with a silicon-based side chain to form an allylsilane, a well-understood functional group. The researchers showed that the allylsilane activates the molecule to react with a suitable trifluoromethyl source, a copper-activated form of a molecule known Togni's reagent. This generates a product in which the trifluoromethyl group is attached at a singly bonded carbon.
"To date, these trifluoromethylated compounds have been difficult to make—our reactions provide a general and efficient method for constructing these types of compounds," Sodeoka says. "We hope that these compounds will contribute to the creation of new trifluoromethylated pharmaceutical drugs."
The process is versatile because the silicon group can be either eliminated or retained during the reaction, depending on the structure of the starting molecule. Leaving the silicon in place offers a way to further functionalize the molecule at that position, if required.
The team is currently investigating the reaction's exact mechanism, particularly regarding the trifluoromethyl source. Only certain copper species work, the researchers found, and the reasons why remain unclear.
Sodeoka and colleagues are also looking to push into new areas of trifluoromethylation chemistry. "We want to develop new types of trifluoromethylation reaction that could provide more complex and useful trifluoromethylated building blocks," she says.
More information: Shimizu, R., Egami, H., Hamashima, Y. & Sodeoka, M. Copper-catalyzed trifluoromethylation of allylsilanes. Angewandte Chemie International Edition 51, 4577–4580 (2012). dx.doi.org/10.1002/anie.201201095
Journal information: Angewandte Chemie International Edition
Provided by RIKEN