Why plants don't get sunburn
Plants rely on sunlight to make their food, but they also need protection from its harmful rays, just like humans do. Recently, scientists discovered a group of molecules in plants that shields them from sun damage. Now, in an article in the Journal of the American Chemical Society, one team reports on the mechanics of how these natural plant sunscreens work.
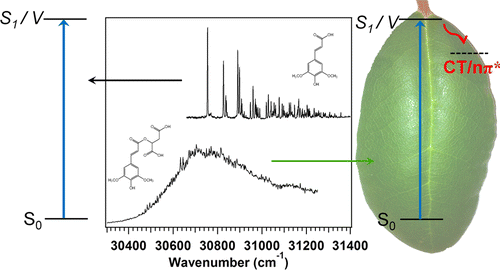
Timothy Zwier and colleagues at Purdue University note that the harsh ultraviolet radiation plants are exposed to daily can cause serious damage to plant DNA and, as a result, hinder plant growth. Biochemical tests have shown that plants produce special molecules and send them to the outer layer of their leaves to protect themselves. These molecules, called sinapate esters, appear to block ultraviolet-B radiation from penetrating deeper into leaves where it might otherwise disrupt a plant's normal development. Although researchers have been amassing evidence that points to sinapate esters as the protective molecules, no one had investigated in detail what happens to them under UV exposure. Zwier's team wanted to understand this process.
The researchers coaxed these molecules into the gas phase and zapped them with UVB radiation from a laser in the laboratory. They found that the particular sinapate ester that plants use as a screen against UVB was inherently capable of soaking up radiation at every wavelength across the UVB spectrum. Thus, it is remarkably efficient at absorbing harsh radiation that could otherwise damage the plant. Their findings further shore up the idea that this class of molecules does indeed comprise plant-made sunblock, the researchers say.
More information: "Plant Sunscreens in the UV-B: Ultraviolet Spectroscopy of Jet-Cooled Sinapoyl Malate, Sinapic Acid, and Sinapate Ester Derivatives" J. Am. Chem. Soc., 2014, 136 (42), pp 14780–14795. DOI: 10.1021/ja5059026
Abstract
Ultraviolet spectroscopy of sinapoyl malate, an essential UV-B screening agent in plants, was carried out in the cold, isolated environment of a supersonic expansion to explore its intrinsic UV spectral properties in detail. Despite these conditions, sinapoyl malate displays anomalous spectral broadening extending well over 1000 cm–1 in the UV-B region, presenting the tantalizing prospect that nature's selection of UV-B sunscreen is based in part on the inherent quantum mechanical features of its excited states. Jet-cooling provides an ideal setting in which to explore this topic, where complications from intermolecular interactions are eliminated. In order to better understand the structural causes of this behavior, the UV spectroscopy of a series of sinapate esters was undertaken and compared with ab initio calculations, starting with the simplest sinapate chromophore sinapic acid, and building up the ester side chain to sinapoyl malate. This "deconstruction" approach provided insight into the active mechanism intrinsic to sinapoyl malate, which is tentatively attributed to mixing of the bright V (1ππ*) state with an adiabatically lower 1nπ* state which, according to calculations, shows unique charge-transfer characteristics brought on by the electron-rich malate side chain. All members of the series absorb strongly in the UV-B region, but significant differences emerge in the appearance of the spectrum among the series, with derivatives most closely associated with sinapoyl malate showing characteristic broadening even under jet-cooled conditions. The long vibronic progressions, conformational distribution, and large oscillator strength of the V (ππ*) transition in sinapates makes them ideal candidates for their role as UV-B screening agents in plants.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society