Finding supports model on cause of DNA's right-handed double helix
The DNA of every organism on Earth is a right-handed double helix, but why that would be has puzzled scientists since not long after Francis Crick and James Watson announced the discovery of DNA's double-helical structure in 1953.
It's a puzzle because no one has been able to think of a fundamental reason why DNA couldn't also be left-handed.
New research by University of Nebraska-Lincoln physicists and published in the Sept. 12 online edition of Physical Review Letters now gives support to a long-posited but never-proven hypothesis that electrons in cosmic rays—which are mostly left-handed—preferentially destroyed left-handed precursors of DNA on the primordial Earth.
The hypothesis, called the Vester-Ulbricht model, was proposed by Frederic Vester of the University of Saarbrucken in Germany and Tilo L.V. Ulbricht of the University of Cambridge in England in 1961 in response to the 1957 discovery that most of the electrons spewing from radioactive beta decay were left-handed.
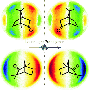
Joan M. Dreiling and Timothy J. Gay of UNL focused circularly polarized laser light on a specially prepared crystal of gallium-arsenide to produce electrons whose spins were either parallel or anti-parallel to their direction of motion upon emission from the crystal—essentially artificial beta rays. They then directed these electrons to strike target molecules of a substance called bromocamphor, which comes in both right- and left-handed varieties.
They found that at the lowest electron energies they studied, left-handed electrons preferentially destroyed left-handed molecules and vice versa. This sensitivity to molecular handedness has a mechanical analog: the inability of a left-handed bolt to screw into a right-handed nut. The molecular experiment proves the principle underlying the Vester-Ulbricht hypothesis.

"The circular polarization of the laser light effectively transferred to the spin (handedness) of the electrons emitted by the gallium-arsenide crystal," said Dreiling, a postdoctoral research assistant who received her doctorate from UNL in May. "We are able to reverse the spin-polarization of the electrons just by reversing the circular polarization of the light."
The effect they saw was quite small, they said—like "looking for an electronic needle in a haystack," Gay said—but they said they're highly confident in their result.
"We have done several different checks with our experiment and I am totally confident that the asymmetry exists," Dreiling said. "The checks all came out showing that this asymmetry is real."
Gay, a professor of physics and astronomy, said the paper in Physical Review Letters culminates a 21-year effort that began in earnest when he came to UNL from the University of Missouri-Rolla in 1993.
"This has been an incredibly hard experiment," he said. "I've ground two graduate students in the dust. Poor Joan survived. The others got their Ph.D.s in other things and a lot of good science came out on the way, but Joan was clever enough to make this experiment work.
"What she did was make the first experiment that showed the asymmetry at the molecular, nano level. That's the molecular physics part of it, which is what we're really interested in, but there's also this tie to the origins of life on Earth."
More information: Chirally Sensitive Electron-Induced Molecular Breakup and the Vester-Ulbricht Hypothesis, journals.aps.org/prl/abstract/ … ysRevLett.113.118103
Journal information: Physical Review Letters
Provided by University of Nebraska-Lincoln