Nanoscale velcro used for molecule transport
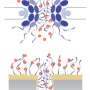
Biological membranes are like a guarded border. They separate the cell from the environment and at the same time control the import and export of molecules. The nuclear membrane can be crossed via many tiny pores. Scientists at the Biozentrum and the Swiss Nanoscience Institute at the University of Basel, together with an international team of researchers, have discovered that proteins found within the nuclear pore function similar to a velcro. In Nature Nanotechnology, they report how these proteins can be used for controlled and selective transport of particles.
There is much traffic in our cells. Many proteins, for example, need to travel from their production site in the cytoplasm to the nucleus, where they are used to read genetic information. Pores in the nuclear membrane enable their transport into and out of the cell nucleus. The Argovia Professor Roderick Lim, from the Biozentrum and the Swiss Nanoscience Institute at the University of Basel, studies the biophysical basics of this transport. In order to better understand this process, he has created an artificial model of the nuclear pore complex, together with scientists from Lausanne and Cambridge, which has led to the discovery that its proteins function like a nanoscale "velcro" which can be used to transport tiniest particles.
"Dirty velcro" inside the nuclear pore
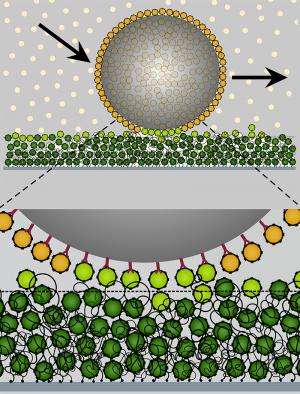
Nuclear pores are protein complexes within the nuclear membrane that enables molecular exchange between the cytoplasm and nucleus. The driving force is diffusion. Nuclear pores are lined with "velcro" like proteins. Only molecules specially marked with import proteins can bind to these proteins and thus pass the pore. But for all non-binding molecules the nuclear pore acts as a barrier. The researchers postulated that transport depends on the strength of binding to the "velcro" like proteins. The binding should be just strong enough that molecules to be transported can bind but at the same time not too tight so that they can still diffuse through the pore.
In an artificial system recreating the nuclear pore, the researchers tested their hypothesis. They coated particles with import proteins and studied their behavior on the molecular "velcro". Interestingly, the researchers found parallels in behavior to the velcro strip as we know it. On "clean velcro", the particles stick immediately. However, when the "velcro" is filled or "dirtied" with import proteins, it is less adhesive and the particles begin to slide over its surface just by diffusion. "Understanding how the transport process functions in the nuclear pore complex was decisive for our discovery," says Lim. "With the nanoscale 'velcro' we should be able to define the path to be taken as well as speed up the transport of selected particles without requiring external energy."
Potential lab-on-a-chip technology applications
Lim's investigations of biomolecular transport processes form the basis for the discovery of this remarkable phenomenon that particles can be transported selectively with a molecular "velcro". "This principle could find very practical applications, for instance as nanoscale conveyor belts, escalators or tracks," explains Lim. This could also potentially be applied to further miniaturize lab-on-chip technology, tiny labs on chips, where this newly discovered method of transportation would make today's complex pump and valve systems obsolete.
More information: Kai D. Schleicher, Simon L. Dettmer, Larisa E. Kapinos, Stefan Pagliara, Ulrich F. Keyser, Sylvia Jeney and Roderick Y.H. Lim. "Selective Transport Control on Molecular Velcro made from Intrinsically Disordered Proteins." Nature Nanotechnology; published online 15 June 2014 | DOI: 10.1038/nnano.2014.103
Journal information: Nature Nanotechnology
Provided by University of Basel