An answer to the perennial question: Is it safe to pee in the pool?
Sanitary-minded pool-goers who preach "no peeing in the pool," despite ordinary and Olympic swimmers admitting to the practice, now have scientific evidence to back up their concern. Researchers are reporting that when mixed, urine and chlorine can form substances that can cause potential health problems. Their study appears in ACS' journal Environmental Science & Technology.
Jing Li, Ernest Blatchley, III, and colleagues note that adding chlorine to pool water is the most common way to kill disease-causing microbes and prevent swimmers from getting sick. But as people swim, splash, play—and pee—in the pool, chlorine mixes with sweat and urine and makes other substances. Two of these compounds, including trichloramine (NCl3) and cyanogen chloride (CNCl), are ubiquitous in swimming pools. The first one is associated with lung problems, and the second one can also affect the lungs, as well as the heart and central nervous system. But scientists have not yet identified all of the specific ingredients in sweat and urine that could cause these potentially harmful compounds to form. So Li's team looked at how chlorine interacts with uric acid, a component of sweat and urine.
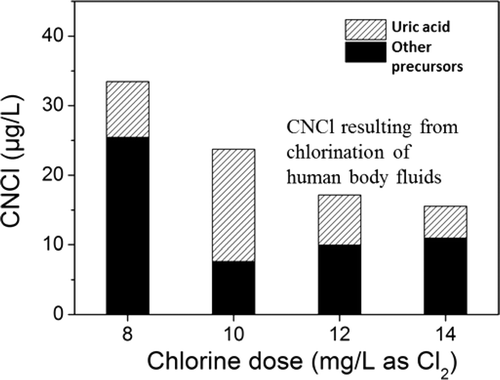
They mixed uric acid and chlorine, and within an hour, both NCl3 and CNCl formed. Though some uric acid comes from sweat, the scientists calculated that more than 90 percent of the compound in pools comes from urine. They conclude that swimmers can improve pool conditions by simply urinating where they're supposed to—in the bathrooms.
More information: "Volatile Disinfection Byproducts Resulting from Chlorination of Uric Acid: Implications for Swimming Pools" Environ. Sci. Technol., 2014, 48 (6), pp 3210–3217. DOI: 10.1021/es405402r
Abstract
Cyanogen chloride (CNCl) and trichloramine (NCl3) are important disinfection byproducts in chlorinated swimming pools. However, some unknowns exist regarding the precursors of their formation. In this study, uric acid is shown to be an efficient precursor to formation of CNCl and NCl3. The molar yields of CNCl and NCl3 were observed to be as high as 44% (pH = 6.0, chlorine/precursor molar ratio [Cl/P] = 6.4) and 108% (pH = 7.0, Cl/P = 30), respectively, both being strong functions of Cl/P, pH, and temperature. Analysis of swimming pool water samples, combined with the results of experiments involving chlorination of uric acid, and chlorination of body fluid analog mixtures, indicated that uric acid chlorination may account for a large fraction of CNCl formation in swimming pools. Moreover, given that uric acid introduction to pools is attributable to urination, a voluntary action for most swimmers, these findings indicate important benefits to pool water and air chemistry that could result from improved hygiene habits on the part of swimmers.
Journal information: Environmental Science & Technology
Provided by American Chemical Society














