A major hub for cell-fate decisions
In a recently published study, LMU researchers show that, in a nerve-cell lineage in the nematode Caenorhabditis elegans, a single protein controls the rate of cell-cycle progression, and decides whether cells divide, differentiate or die.
The members of a proliferating cell lineage repeatedly face a choice between continued cell division, the adoption of a terminally differentiated state, and programmed cell death. In the interests of the organism as a whole, these processes must be carefully coordinated but, in many cases, the mechanisms that enable individual cells to choose one or the other remain obscure. LMU developmental biologist Professor Barbara Conradt and her team have now found that, in the model organism Caenorhabditis elegans, all three processes are regulated by the protein CES-1, and determined how it influences cell-fate choice.
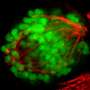
"The point of departure for our study was the question of how programmed cell death is regulated in the so-called NSM cell lineage in C. elegans," says Conradt, who is also a member of the Center for Integrated Protein Science Munich (CIPSM), a Cluster of Excellence. In this nerve-cell lineage, a stem cell or neuroblast, divides to give rise to two daughter cells of unequal size. The smaller cell dies within 30 minutes after the completion of division, while the other differentiates into a specific type of neuron. "We had previously shown that CES-1 can inhibit the death of the smaller daughter. The protein is normally found in significant amounts only in the larger daughter – but if its level in the smaller cell is artificially boosted, that cell also survives. In addition, CES-1 also plays a role in determining how the mother cell divides. In mutants that overproduce CES-1, the neuroblast divides symmetrically to generate daughters of equal size, both of which survive. This tells us that CES-1 has an influence on the polarity of the mother cell – which is defined by a specific spatial distribution of certain components in the cell, which then determines the position of the plane of division.
Using a combination of genetic and cell biological methods, the researchers have now shown that, in addition to its influence on cell polarity and programmed cell death, CES-1 also regulates another basic aspect of the mother cell's behavior. "We believe that CES-1 also determines the length of time that must elapse before it divides – in other words, the duration of the cell cycle. The NSM neuroblast divides about two and a half hours after its formation, and CES-1 accomplishes its diverse functions within this short period of time," Conradt says.
CES-1 belongs to the Snail-like family of transcription factors, and Snail-like proteins have previously been implicated in the control of cell polarity, cell division and programmed cell death in a range of experimental systems. "However, no one has ever shown before that all three processes can be regulated in rapid succession by one and the same Snail-like protein. So CES-1 is an immensely important switch in the circuitry that controls cell division and cell fate," says Conradt. This multifaceted function probably explains why Snail-like factors have been found to play a significant role in tumorigenesis and, in particular, in the process of metastasis. CES-1 is therefore a very interesting target for the development of new therapeutic agents for the treatment of cancer.
Conradt and her team will now construct a genome-wide map of the chromosomal regions to which CES-1 binds. By employing a systematic mapping approach, they hope to elucidate the mechanism by which CES-1 alters the polarity of the NSM neuroblast. CES-1 is known to bind to many sites in the genome and regulates perhaps as many as 3000 genes. "Many of these will undoubtedly turn out to be genes with very exciting functions that have not previously been identified as targets for Snail-like proteins. So we have plenty to do!", Conradt concludes.
More information: Yan B, Memar N, Gallinger J, Conradt B (2013) Coordination of Cell Proliferation and Cell Fate Determination by CES-1 Snail. PLoS Genet 9(10): e1003884. DOI: 10.1371/journal.pgen.1003884
Journal information: PLoS Genetics
Provided by Ludwig Maximilian University of Munich