New aluminum alloy stores hydrogen
We use aluminum to make planes lightweight, store sodas in recyclable containers, keep the walls of our homes energy efficient and ensure that the Thanksgiving turkey is cooked to perfection. Now, thanks to a group of Japanese researchers, there may soon be a new application for the versatile metal: hydrogen storage for fuel cells.
Lightweight interstitial hydrides—compounds in which hydrogen atoms occupy the interstices (spaces) between metal atoms—have been proposed as a safe and efficient means for storing hydrogen for fuel cell vehicles. Hydrides using magnesium, sodium and boron have been manufactured, but so far, none have proven practical as a hydrogen repository. An aluminum-based alloy hydride offers a more viable candidate because it has the desired traits of light weight, no toxicity to plants and animals, and absence of volatile gas products except for hydrogen. Until now, however, only complex aluminum hydrides—unsuitable for use as a hydrogen storage system—have been created.
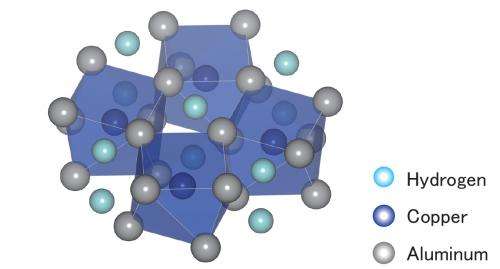
In a recent paper in the AIP Publishing journal APL Materials, a joint research group with members from the Japan Atomic Energy Agency (Hyogo, Japan) and Tohoku University (Sendai, Japan) announced that it had achieved the long-sought goal of a simple-structured, aluminum-based interstitial alloy. Their compound, Al2CuHx, was synthesized by hydrogenating Al2Cu at an extreme pressure of 10 gigapascals (1.5 million pounds per square inch) and a high temperature of 800 degrees Celsius (1,500 degrees Fahrenheit).
The researchers characterized the conditions of the hydrogenation reaction using in-situ synchrotron radiation X-ray diffraction measurement, while the crystal and electron structures of the compound formed were studied with powder X-ray diffraction measurement and first-principle calculations, respectively. Together, these examinations confirmed the first-ever formation of an interstitial hydride of an aluminum-based alloy.

"Although its synthesis requires very extreme conditions and its hydrogen content is low, our new compound showed that an aluminum-based alloy hydride is achievable," said Hiroyuki Saitoh, lead author of the APL Materials paper.
"Based on what we've learned from this first step, we plan to synthesize similar materials at more moderate conditions—products that hopefully will prove to be very effective at storing hydrogen."
More information: The article, "Synthesis and formation process of Al2CuHx: A new class of interstitial aluminum-based alloy hydride" is authored by Hiroyuki Saitoh, Shigeyuki Takagi, Naruki Endo, Akihiko Machida, Katsutoshi Aoki, Shin-ichi Orimo and Yoshinori Katayama. It appears in the journal APL Materials: dx.doi.org/10.1063/1.4821632
Provided by American Institute of Physics