New detection technique makes flash photos of proteins
Researchers from FOM research institute AMOLF in the Netherlands have produced the first images of a cloud of heavy protein ions during deposition and ionization. The images, enabled by an innovative detector, have provided the researchers with an insight into how protein ions on tissue are formed. The research has been published online on 3 September 2013 as a Very Important Paper (VIP) in Angewandte Chemie.
To produce the images, researchers from the group of professor Ron Heeren combined the so-called Timepix detector with a mass spectrometer, conventional detectors struggle with demonstrating the presence of large intact proteins in biological samples.
Well combined
The Timepix detector was developed for high-energy physics, so the AMOLF researchers first had to adapt it for protein analysis. They did this by removing a photon sensor and replacing it with a plate sensitive for charged particles such as ions. The Timepix detector chip consists of 262,144 individual pixels, each of which separately measures the mass and location of the protein ions. Consequently the detector does not make a single measurement but produces 262,144 spectra from a single desorption event. The information density therefore strongly increases.
The researchers connected the Timepix detector to a time of flight mass spectrometer. Such a spectrometer accelerates laser produced protein ions with the help of an electric field. Ions with the same charge receive the same amount of kinetic energy, but their actual speed is dependent on their mass. The lighter the particle, the sooner it reaches the detector. With this approach the mass-to-charge ratio of the molecules can be determined. This ratio is unique for each molecule as a result of which the device can precisely determine which protein comes into contact with the detector.
Using the Timepix method the researchers can detect heavy molecules with a greater speed and sensitivity than is usually the case. Furthermore, the Timepix also determines the position of the molecule at the moment this comes into contact with the detector, something that a conventional mass spectrometer cannot do.
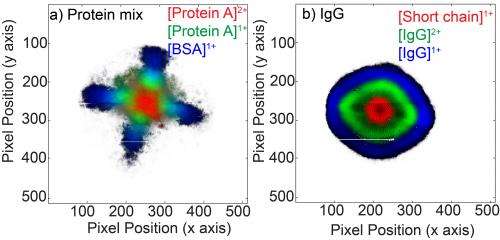
Ions imaged
Using this system the researchers investigated the heavy antibody-protein immunoglobulin G. They saw a series of ions that are not or are scarcely visible with a standard detector. The researchers also imaged the position of the various molecules. That way, they could make time-lapse images every 50 nanoseconds of the ion cloud at the moment it touched the detector surface.
These are the first images to demonstrate the laser-driven formation process of the protein ions. The images also show how the lenses of the mass spectrometer influence the image. With the images the researchers have obtained a unique insight into the fundamental physics that occurs in the ion source. They can now put this information to immediate use to optimise the performance of the entire mass spectrometric system.
"The advantage of the Timepix detector is that besides being able to measure more than 100 times more sensitively, you can also use it to make detailed images", says Ron Heeren. "With the detector, molecular flash photography has now become a mature technique."
Journal information: Angewandte Chemie




















