Tumors elude anti-cancer drugs through 'fork reversal' repair
In research recently published in Nature Structural & Molecular Biology, Alessandro Vindigni, Ph.D., associate professor of biochemistry and molecular biology at Saint Louis University, discovered how cancer cells respond to the damage caused by an important class of anti-cancer drugs, topoisomerase I inhibitors. The discovery points to opportunities to improve chemotherapeutic regimens based on topoisomerase I inhibitor treatment and limit their toxic side effects.
"Most cancer chemotherapeutics act by inhibiting DNA replication," Vindigni said. "The drugs aim to target highly proliferating cancer cells rather than normal cells, but unfortunately many also are toxic for normal cells."
Cells, both healthy and cancerous, reproduce by replicating their DNA. Anti-cancer drugs like topoisomerase I (TOP1) inhibitors work by disrupting DNA replication in cancer cells.
As a cell's double helix-shaped DNA strands split apart and begin to copy themselves, the long strands get wound up, like a rope that's been twisted too many times. The coils must be released or replication can't continue to move forward. Topoisomerase is an enzyme that temporarily cuts the rope of DNA, lets it untwist, and puts it back together again. If you block this process with a TOP1 inhibitor, the rope remains knotted and cell duplication stops.
Scientists' understanding of how TOP1 inhibitors work has, however, been incomplete. The working theory has been that TOP1 inhibitors leave a nick on one of the DNA strands by inhibiting the ability of the topoisomerase to seal the broken DNA. When the replication machinery collides with this nick, a double strand break occurs which stops the replication process. Double strand breaks are the worst kind of DNA lesions because both strands are compromised and require a complicate DNA repair pathway to be fixed.
In his recent findings, however, Vindigni and his team found that the cells are much "smarter" than they originally appeared.
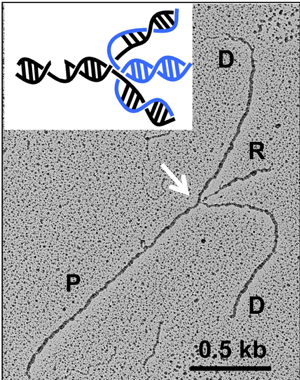
Living up to their crafty reputation, cancer cells have a strategy to deal with this scenario. When TOP1 inhibition causes "replication stress" in the form of a nick on the DNA, the replication machinery pauses and reverses its course instead of colliding with the TOP1 induced DNA lesion. This mechanism of "replication fork reversal" gives time for the lesion to be repaired, so that replication can continue on again, preventing the hoped-for double strand break.
In fact, not only does the strand halt when it reaches the nick, but it senses the problem ahead of time, coming down the line. The replication structure does not operate blindly, but has advance notice of the injury, which gives it time to halt and repair the lesion before it reverses course and move into forward gear again.
"This is important because it is the first evidence that says that cancer cells' DNA replication machinery can react to drug treatment through fork reversal. Now we also uncovered the mechanism to restart the replication forks."
Vindigni's team also found that two important cellular proteins, PARP and RECQ1, control the fork reversal mechanism.
In particular, RECQ1, which is an enzyme that plays a key role in the maintenance of genome stability, appears to be responsible for restarting the reversed replication forks once the TOP1 induced lesion has been repaired. The reversed forks cannot restart without the help of RECQ1, suggesting it may be a promising target for a drug therapy.
Once they undercut the replication fork reversal process, researchers can return to the approach of encouraging a "fatal" double strand break for cancer cells by combing TOP1 inhibitors with novel inhibitors of the proteins that control the process of replication fork reversal and restart. In addition, inducing replication fork reversal by TOP1 inhibitor treatment and impeding reversed replication fork restart by RECQ1 inhibition should also stop DNA replication, thus allowing doctors to use lower TOP1 inhibitor doses, which, in turn, would mean fewer side effects.
What's next?
Researchers' next step is to determine if this mechanism holds true not just in response to TOP1 inhibitors, but also with other cancer drugs, a finding that would broaden this paper's significance even further.
Another important step will be to search for other factors that control the replication fork reversal process, findings which would offer additional opportunities to disrupt the process.
"TOP1 drugs are widely used in clinic for many types of cancer. However, they also are highly toxic," Vindigni said. "We discovered the mechanism that the cancer cells' replication machinery uses to respond to treatment with these drugs. We also discovered the factors that control this mechanism.
"We hope to combine new RECQ1 inhibitors with existing drugs to create more effective and specific therapies with fewer toxic side effects."
More information: www.nature.com/nsmb/journal/v2 … /full/nsmb.2501.html
Journal information: Nature Structural & Molecular Biology
Provided by Saint Louis University














