New catalyst dives into water to produce hydrogen

(Phys.org) —Few catalysts are energy efficient, highly active, stable, and operate in water, but a nickel-based catalyst designed at the Center for Molecular Electrocatalysis at Pacific Northwest National Laboratory quickly produces hydrogen molecules in solutions with 75 percent water. This catalyst contains tailored relays that allow the catalyst to quickly shuttle protons from the solution to the heart of the catalyst, where they are added to electrons. The catalyst is known to be energy efficient, stable and highly active. With the modified design, it now operates in water, producing up to 170,000 hydrogen molecules per second. The study on this catalyst was highlighted as a hot article in Chemical Communications.
"We've moved from pure organic solvents to solutions with increasing amounts of water," said Dr. Monte Helm, Deputy Director of the Center for Molecular Electrocatalysis, an Energy Frontier Research Center. "We found that our catalyst performed better with water than in an organic solvent alone."
Solar power is not a large part of our nation's energy grid, in part because of its intermittent nature and the challenges involved in storing the energy. One option is to combine the electrons generated with protons and create molecular hydrogen. The hydrogen stores the energy for later use. Catalysts are needed to drive this reaction, and this study answers a fundamental question about these catalysts by showing how to design proton relays that achieve fast, energy-efficient materials able to work in water.
"We are getting closer to what we need for a real-world catalyst," said Dr. Morris Bullock, Director of the Center for Molecular Electrocatalysis.
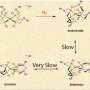
The team began with a stable, efficient nickel-based catalyst. The catalyst is [Ni(PPh2NC6H4OH2)2](BF4)2, where PPh2NC6H4OH2 equals 1,5-bis(p-hydroxyphenyl)-3,7-diphenyl-1,5-diaza-3,7-diphosphacyclo-octane. The catalyst drives a reaction that produces hydrogen by adding protons and electrons.
The p-hydroxyphenyl groups in the catalyst's structure make it hydrophilic, able to easily mix with water. They synthesized the catalyst in the quantities needed for experiments and determined the maximum amount of water that the catalyst could tolerate. They found that the catalyst would work in a solution with up to 75 percent water.
The scientists conducted a host of electrochemical hydrogen production studies. The energy efficiency or overpotential was determined to be 310 to 470 millivolts with turnover rates of up to 170,000/second, one of the fastest and most efficient of its kind.
Further, the team determined the catalyst's behavior in the presence of different amounts of acid. The acid supplies the protons that are relayed via pendant amines to the catalyst's active site. The team found that there is an optimum pH where the catalyst performs best.
"This work underscores the important roles that the pendent amine, acid and solvent medium all play in determining the catalyst's performance," said Dr. John Roberts, a scientist at the EFRC.
The team continues to uncover the basic design principles for catalysts capable of hydrogen oxidation and hydrogen production.
More information: Hoffert WA, JAS Roberts, RM Bullock, and ML Helm. 2013. Production of H2 at Fast Rates using a Nickel Electrocatalyst in Water-Acetonitrile Solutions, Chemical Communications 49:7767-7769. DOI: 10.1039/C3CC43203C
Journal information: Chemical Communications
Provided by Pacific Northwest National Laboratory