Crystal structure reveals light regulation in cyanobacteria
Light is crucial for photosynthetic organisms, but one can have too much of a good thing. Excess light can harm organisms when the amount of energy absorbed exceeds the rate of carbon fixation.
To protect themselves from excess light, cyanobacteria rely on light- harvesting antennae called phycobilisomes that can sense light conditions in order to efficiently collect this energy. A protein known as OCP helps regulate the phycobilisomes, and is in turn regulated by a protein known as FRP.
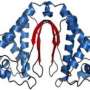
In an article published ahead online May 28, 2013 in Proceedings of the National Academy of Sciences, a team led by DOE Joint Genome Institute researchers focused on the crystal structure of the FRP protein in a species of Synechocystis bacteria to learn more about how cyanobacteria protect themselves from excess light.
They were able to look at the crystal structure using beamlines at Berkeley Lab's Advanced Light Source. They found that FRP has two forms: it forms a dimer in the active state and otherwise appears as a tetramer. The researchers also found a region of highly conserved residues on the dimer structure of FRP that are essential to the protein's activity.
Their findings have led the team to propose a model of how the FRP and OCP proteins interact with phycobilisomes under both high light and low light conditions in order to ensure the cyanobacteria can safely and efficiently harness light to conduct photosynthesis.
More information: www.pnas.org/content/early/201 … /1303673110.abstract
Journal information: Proceedings of the National Academy of Sciences
Provided by US Department of Energy