Monitoring interactions between proteins in their natural environment with intracellular labeling technique
One can often begin to understand a protein's function by identifying its accomplices in the cell. Accordingly, scientists have developed diverse biochemical assays that essentially allow them to go 'fishing' for interaction data—using a purified protein of interest as 'bait' to pluck its binding partners from out of a cellular extract.
Such assays are not always an option, however; many cellular proteins are very difficult to purify, making it impractical to use them as bait in a binding assay. For example, Kyle Roux of the Sanford Children's Health Research Center, USA, has encountered persistent difficulties in his efforts to study interactions with proteins that contribute to the envelope surrounding the cellular nucleus. In collaboration with A*STAR researchers Manfred Raida of the Experimental Therapeutics Centre and Brian Burke of the Institute of Molecular Biology, Roux has now developed a promising solution for dealing with such tricky targets.
They made use of a mutant variation of BirA, a bacterial enzyme that tags nearby proteins with a molecule called biotin. The mutant, BirA, is indiscriminate in its labeling. Roux and co-workers predicted that any cellular protein that gets fused to BirA should permanently mark its interacting partners with biotin. This would make them easy to isolate after the assay is done. Most importantly, these experiments can be done in the milieu of a living cell, making them more 'natural' than conventional binding assays.
As a test case, the researchers used lamin A, a nuclear envelope-associated protein that confounds efforts at purification by forming insoluble clumps. Their technique, which they termed BioID, proved highly effective, yielding more than 120 biotin-marked proteins that potentially interacted with lamin A over the course of the 24-hour labeling period. Even fleeting associations can result in BirA labeling, notes Roux. "BioID has the potential to capture those weak and transient interactions that the other biochemical methods often miss," he says, but adds that follow-up experimental confirmation will still be required.
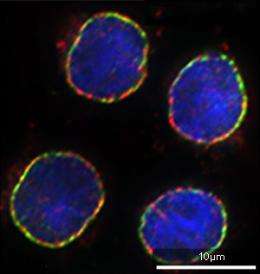
The team's demonstration of BioID also netted at least one previously uncharacterized protein that appears to represent a bona fide nuclear envelope constituent (see image). Roux is confident that this technique could prove a potent tool for mapping interaction networks in structures throughout the cell. "We have already successfully applied BioID to integral membrane proteins," he says. "We have also used BioID in the mitochondrial matrix and at the nuclear pore complex, and these studies will soon expand to the extracellular space."
More information: Roux, J. K., Kim, D. I., Raida, M. & Burke, B. A promiscuous biotin ligase fusion protein identifies proximal and interacting proteins in mammalian cells. Journal of Cell Biology 196, 801–810 (2012). jcb.rupress.org/content/early/ … 6/jcb.201112098.full
Journal information: Journal of Cell Biology













