Scientists show how cells accurately inherit information that is not contained in their genes
All 10 trillion cells in the adult human body are genetically identical, but develop into distinct cell types, such as muscle cells, skin cells or neurons, by activating some genes while inhibiting others. Remarkably, each specialized cell maintains a memory of their individual identity by remembering which genes should be kept on or off, even when making copies of themselves. This type of memory is not written directly into the DNA, yet it is heritable. Instead, such non-genetic or "epigenetic" instructions often appear to be contained in proteins and control not only genes but also how chromosomes are organized.
Lars Jansen and his team, at the Instituto Gulbenkian de Ciência (IGC), have worked out how one of these epigenetic organizing centres is faithfully passed on from mother to daughter cells. Their findings not only elucidate a hitherto mystifying biological process, but provide insight into a key process in cell division, one that can cause cancer when it goes wrong. These findings feature in the latest issue of the journal Developmental Cell.
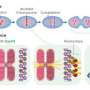
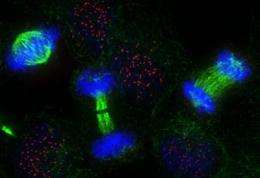
The team focused on the centromere, a unique protein structure on each chromosome that attaches it to the cell's skeleton (cytoskeleton) during cell division and thus ensures that each daughter cell receives exactly one set of freshly made chromosomes. A properly functioning centromere is critical, as loss in accuracy of this process results in cells with the wrong number of genes, a hallmark of tumor cells.
Mariana Silva, a PhD student in the lab, and first author of the study, explains "When cells divide they make exactly two copies of all genes, to be passed on to exactly two cells. A similar feat has to be pulled off for non-genetic information. But how does the cell copy a protein structure? And, how does it ensure just the right number of copies are made? This question is still mystifying scientists. We focussed our efforts on the centromere because the key protein responsible for its epigenetic behavior is known."
This protein, called CENP-A, keeps a "molecular memory" of the centromere, ensuring its inheritance. Previous studies, by Lars and other research groups, had shown that, while cells duplicate their DNA before mitosis, duplication of the centromere, led by the CENP-A protein, takes place only after mitosis (during a 'gap' phase called G1). What triggers its duplication and how accuracy is ensured remained unknown.
Lars and his team now show that the very same machinery that is controlling the well-known process of DNA duplication is also controlling CENP-A duplication. This machinery, that includes the so-called cyclin-dependent kinases (Cdks) acts like a molecular clock driving the different steps of the cell cycle forward, one after the other. When Cdks are highly active (just before mitosis) DNA duplicates while duplication of CENP-A is inhibited. Conversely, when they are inactive (after mitosis), CENP-A is duplicated but at that point DNA duplication is inhibited. In other words, when DNA duplicates at midnight, Cdks make sure that the centromere is copied only at noon.
The IGC researchers came to this elegant model by painstainkingly inhibiting Cdk activity in human and chicken cells at set times. When doing so, they could fool the cells into making new centromeres even while the cells were in the middle of duplicating their DNA. "It´s like a cell with a jetlag", says Lars Jansen.
Lars puts their findings into context "What we've uncovered is a very simple, neat mechanism whereby the cell couples DNA duplication, cell division and centromere assembly. By using the same machinery (Cdks) for all these steps but in opposite ways, the cell makes sure that the right number of copies of both genes and centromeres are made by allowing each the appropriate time. Keeping these critical processes separate in time might be important to avoid errors in either one. Understanding these general principles of epigenetic inheritance are fundamental to our understanding of how genes are regulated, how genomes are organised, and the wide spectrum of diseases that result from errors in these mechanisms".
Provided by Instituto Gulbenkian de Ciencia