A dance of aerosols: Study shows plant-sourced particles pick and choose their partners
(PhysOrg.com) -- In the complex molecular mixer that is the atmosphere, some molecules dance with others and some are wallflowers, according to scientists at Pacific Northwest National Laboratory. Using a unique environmental testing chamber located at PNNL, the team was able to watch vapors and particles mix in real time. The scientists focused on how a specific vapor, responsible for the aromatic scent of fir trees, forms carbon-containing particles that mix with certain carbon-containing acids in the atmosphere. Their findings show that climate models are underestimating the total amount of carbon-containing particles formed in the air.
To arrive at regional and global climate projections, scientists must consider thousands of complex variables to simulate climate processes. One of those complex interactions is aerosols, the microscopic particles of dust, soot, and chemicals dispersed in the atmosphere that scatter or absorb sunlight and act as seeds for cloud formation. A large amount of aerosols are organic, or carbon-containing, and exist in two forms based on their origin: primary and secondary. This study focuses on secondary organic aerosols, formed in the atmosphere through interactions that are much more difficult to represent in models. New research from PNNL further defines the role of SOA so that scientists can more accurately depict their effect on the climate.
"Inorganic aerosols, like sulfates, nitrates, and ammonium, are well-represented in climate models. However, to make climate models more accurate, we are focused on developing a better understanding of the dynamics of organic aerosols formed from plant-based organic vapors and their interaction with aerosols emitted from human activities," said Dr. Chen Song, a PNNL atmospheric scientist.
Much of Earth's land is covered by plants and trees, representing a huge source of biogenic, or plant-based, organic aerosols. The PNNL study measured how, in the atmosphere, these aerosols interact with and mix with other volatile or semi-volatile organic compounds, the carbon-centric chemicals that evaporate from both natural and human-made sources. Existing climate models assumed that these organic compounds will mix equally with those already in the atmosphere.
Song and fellow researchers published research in 2007 that addressed assumptions in the ability of diesel exhaust organic aerosols to mix with organic aerosols from tree evaporate. This new study, published in the journal Environmental Science & Technology, furthered this research by examining the changes in physical properties of these aerosols due to aging, or oxidation, provoked by ozone or other oxidizing agents.
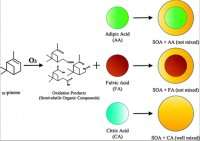
Researchers used PNNL's Environmental Chamber to simulate and study the lifecycle of these aerosols. The team injected citric, adipic, and fulvic acid into the chamber as seed aerosols, and then introduced a secondary organic aerosol from α-pinene, a carbon-containing compound derived from pine trees.
The results showed that organic aerosols from biogenic volatile organic compounds will not mix with all oxidized organic aerosols in the atmosphere, as assumed in existing climate models. Rather, they will only mix with certain, highly oxidized aerosols. According to Song, this finding highlights the need to improve how organic aerosols are currently represented in climate models. When volatile organic compounds oxidize, not all will become aerosols. Some stay in the gas phase and others start a transfer into the aerosol phase. The transformation mechanism, and at what stage and condition they transition to aerosols, is based on vapor pressure, how much mass is in aerosol phase and the mixing states of the aerosol phase.
The research team will next introduce aerosols from diesel exhaust into the PNNL's Environmental Chamber to better understand the interactions of oxidized diesel aerosols and vapor with biogenic aerosols.
More information: Song C, et al. 2011. "Effect of Hydrophilic Organic Seed Aerosols on Secondary Organic Aerosol Formation from Ozonolysis of α-Pinene," Environmental Science & Technology, 45 (17), 7323-7329. DOI:10.1021/es201225c
Journal information: Environmental Science & Technology
Provided by Pacific Northwest National Laboratory


















