October 7, 2011 feature
High-purity hydrogen generated from a single device
(PhysOrg.com) -- There are many ways to generate hydrogen, such as water electrolysis and steam reforming of gas, but the hydrogen produced by these methods tends to be combined with other byproduct and residual gases. For this reason, a second step to purify the hydrogen is usually required after it is produced. Now in a new study, scientists have developed a method for generating hydrogen with a purity of more than 99% within a single membrane, eliminating the need for a separate purification step.
The researchers, Masashi Hattori, Kei Noda, and Kazumi Matsushige, from Kyoto University in Kyoto, Japan, have published their study on the integration of hydrogen production and purification in a recent issue of Applied Physics Letters.
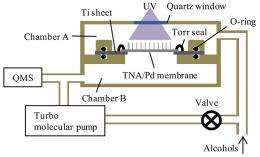
Their method generates hydrogen by shining ultraviolet (UV) light onto a membrane just a few tens of micrometers thick. The membrane is made of two layers: a titanium dioxide nanotube array (TNA), which serves as the photocatalyst to generate the hydrogen, and a palladium (Pd) thin film, which serves as the hydrogen purification part.
The membrane forms the basis of a reactor that also includes a UV light and two chambers, one above and one below the membrane. The researchers pumped a fuel, such as methanol or ethanol, into the upper chamber and then turned on the UV light. The light caused a photocatalytic reaction, turning the fuel into carbon dioxide (CO2), formaldehyde (CH2O), and hydrogen (H2) in the upper chamber. In the lower chamber, after passing through the membrane, the researchers measured the purity of the generated H2 to be 99-100% when using either methanol or ethanol fuel. Although the scientists did detect some small amounts of CO2 and CH2O in the lower chamber, they concluded that these were not byproducts from the hydrogen generation but that they were already present as background gases.
“The purification of the produced hydrogen gas was done with the Pd layer,” Noda told PhysOrg.com. “Only hydrogen can pass through the Pd layer and appear in the lower chamber. Other gases still exist inside the upper chamber.”
By integrating the hydrogen generation and purification processes within a single membrane, the researchers hope that the new device will overcome some of the problems faced by previous approaches. For instance, the small membrane reactor, which operates at room temperature, could lead to the miniaturization and low-energy operation of fuel cells, which could have applications for mobile and on-site hydrogen reforming systems. However, the researchers still have a lot of work to do before the device is ready for these applications.
"The performance of the TNA/Pd membrane we reported here is not satisfactory yet," Noda said. “For example, the amount of produced hydrogen with the membrane is quite low at present. The replacement of Pd with Pd alloys with another metal is also necessary to suppress hydrogen embrittlement. In terms of production cost, the thickness of hydrogen permeable metals should be reduced. But, we will make a lot of effort to improve the performance of this new membrane toward practical applications.”
More information: Masashi Hattori, et al. “High-purity hydrogen generation by ultraviolet illumination with the membrane composed of titanium dioxide nanotube array and Pd layer.” Applied Physics Letters 99, 123107 (2011) DOI:10.1063/1.3643052
Copyright 2011 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.