Research update: New way to store sun's heat
A novel application of carbon nanotubes, developed by MIT researchers, shows promise as an innovative approach to storing solar energy for use whenever it’s needed.
Storing the sun’s heat in chemical form — rather than converting it to electricity or storing the heat itself in a heavily insulated container — has significant advantages, since in principle the chemical material can be stored for long periods of time without losing any of its stored energy. The problem with that approach has been that until now the chemicals needed to perform this conversion and storage either degraded within a few cycles, or included the element ruthenium, which is rare and expensive.
Last year, MIT associate professor Jeffrey Grossman and four co-authors figured out exactly how fulvalene diruthenium — known to scientists as the best chemical for reversibly storing solar energy, since it did not degrade — was able to accomplish this feat. Grossman said at the time that better understanding this process could make it easier to search for other compounds, made of abundant and inexpensive materials, which could be used in the same way.
Now, he and postdoc Alexie Kolpak have succeeded in doing just that. A paper describing their new findings has just been published online in the journal Nano Letters, and will appear in print in a forthcoming issue.
The new material found by Grossman and Kolpak is made using carbon nanotubes, tiny tubular structures of pure carbon, in combination with a compound called azobenzene. The resulting molecules, produced using nanoscale templates to shape and constrain their physical structure, gain “new properties that aren’t available” in the separate materials, says Grossman, the Carl Richard Soderberg Associate Professor of Power Engineering.
Not only is this new chemical system less expensive than the earlier ruthenium-containing compound, but it also is vastly more efficient at storing energy in a given amount of space — about 10,000 times higher in volumetric energy density, Kolpak says — making its energy density comparable to lithium-ion batteries. By using nanofabrication methods, “you can control [the molecules’] interactions, increasing the amount of energy they can store and the length of time for which they can store it — and most importantly, you can control both independently,” she says.
Thermo-chemical storage of solar energy uses a molecule whose structure changes when exposed to sunlight, and can remain stable in that form indefinitely. Then, when nudged by a stimulus — a catalyst, a small temperature change, a flash of light — it can quickly snap back to its other form, releasing its stored energy in a burst of heat. Grossman describes it as creating a rechargeable heat battery with a long shelf life, like a conventional battery.
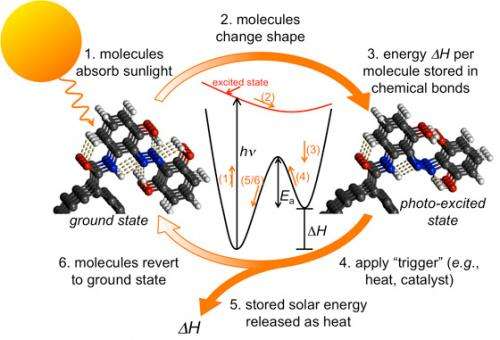
One of the great advantages of the new approach to harnessing solar energy, Grossman says, is that it simplifies the process by combining energy harvesting and storage into a single step. “You’ve got a material that both converts and stores energy,” he says. “It’s robust, it doesn’t degrade, and it’s cheap.” One limitation, however, is that while this process is useful for heating applications, to produce electricity would require another conversion step, using thermoelectric devices or producing steam to run a generator.
While the new work shows the energy-storage capability of a specific type of molecule — azobenzene-functionalized carbon nanotubes — Grossman says the way the material was designed involves “a general concept that can be applied to many new materials.” Many of these have already been synthesized by other researchers for different applications, and would simply need to have their properties fine-tuned for solar thermal storage.
The key to controlling solar thermal storage is an energy barrier separating the two stable states the molecule can adopt; the detailed understanding of that barrier was central to Grossman’s earlier research on fulvalene dirunthenium, accounting for its long-term stability. Too low a barrier, and the molecule would return too easily to its “uncharged” state, failing to store energy for long periods; if the barrier were too high, it would not be able to easily release its energy when needed. “The barrier has to be optimized,” Grossman says.
Already, the team is “very actively looking at a range of new materials,” he says. While they have already identified the one very promising material described in this paper, he says, “I see this as the tip of the iceberg. We’re pretty jazzed up about it.”
Yosuke Kanai, assistant professor of chemistry at the University of North Carolina at Chapel Hill, says “the idea of reversibly storing solar energy in chemical bonds is gaining a lot of attention these days. The novelty of this work is how these authors have shown that the energy density can be significantly increased by using carbon nanotubes as nanoscale templates. This innovative idea also opens up an interesting avenue for tailoring already-known photoactive molecules for solar thermal fuels and storage in general.”
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.
Provided by Massachusetts Institute of Technology