Researchers find a stable way to store the sun's heat (w/ Video)
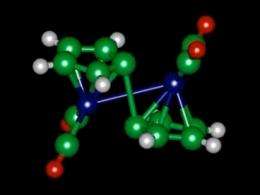
(PhysOrg.com) -- Researchers at MIT have revealed exactly how a molecule called fulvalene diruthenium, which was discovered in 1996, works to store and release heat on demand. This understanding, reported in a paper published on Oct. 20 in the journal Angewandte Chemie, should make it possible to find similar chemicals based on more abundant, less expensive materials than ruthenium, and this could form the basis of a rechargeable battery to store heat rather than electricity.
The molecule undergoes a structural transformation when it absorbs sunlight, putting it into a higher-energy state where it can remain stable indefinitely. Then, triggered by a small addition of heat or a catalyst, it snaps back to its original shape, releasing heat in the process. But the team found that the process is a bit more complicated than that.
"It turns out there's an intermediate step that plays a major role," said Jeffrey Grossman, the Carl Richard Soderberg Associate Professor of Power Engineering in the Department of Materials Science and Engineering. In this intermediate step, the molecule forms a semi-stable configuration partway between the two previously known states. "That was unexpected," he said. The two-step process helps explain why the molecule is so stable, why the process is easily reversible and also why substituting other elements for ruthenium has not worked so far.
In effect, explained Grossman, this process makes it possible to produce a "rechargeable heat battery" that can repeatedly store and release heat gathered from sunlight or other sources. In principle, Grossman said, a fuel made from fulvalene diruthenium, when its stored heat is released, "can get as hot as 200 degrees C, plenty hot enough to heat your home, or even to run an engine to produce electricity."
Compared to other approaches to solar energy, he said, "it takes many of the advantages of solar-thermal energy, but stores the heat in the form of a fuel. It's reversible, and it's stable over a long term. You can use it where you want, on demand. You could put the fuel in the sun, charge it up, then use the heat, and place the same fuel back in the sun to recharge."
In addition to Grossman, the work was carried out by Yosuke Kanai of Lawrence Livermore National Laboratory, Varadharajan Srinivasan of MIT's Department of Materials Science and Engineering, and Steven Meier and Peter Vollhardt of the University of California, Berkeley.
The problem of ruthenium's rarity and cost still remains as "a dealbreaker," Grossman said, but now that the fundamental mechanism of how the molecule works is understood, it should be easier to find other materials that exhibit the same behavior. This molecule "is the wrong material, but it shows it can be done," he said.
The next step, he said, is to use a combination of simulation, chemical intuition, and databases of tens of millions of known molecules to look for other candidates that have structural similarities and might exhibit the same behavior. "It's my firm belief that as we understand what makes this material tick, we'll find that there will be other materials" that will work the same way, Grossman said.
Grossman plans to collaborate with Daniel Nocera, the Henry Dreyfus Professor of Energy and Professor of Chemistry, to tackle such questions, applying the principles learned from this analysis in order to design new, inexpensive materials that exhibit this same reversible process. The tight coupling between computational materials design and experimental synthesis and validation, he said, should further accelerate the discovery of promising new candidate solar thermal fuels.
More information: "Mechanism of Thermal Reversal of the (Fulvalene) tetracarbonyldiruthenium Photoisomerization: Toward Molecular Solar–Thermal Energy Storage," by Yosuke Kanai, Varadharajan Srinivasan, Steven K. Meier, K. Peter C. Vollhardt, Jeffrey C. Grossman. Angewandte Chemie, 20 October, 2010.
Provided by Massachusetts Institute of Technology


















