Twin zinc atoms can direct an important organic double-bond-forming reaction with high efficiency
Many natural compounds found in plant and animals display potent medicinal capabilities, but their intricate chemical structures prevent large-scale manufacturing. One common difficulty is synthesizing carbon–carbon double bonds, or alkenes, on the exterior of a molecular framework—a reactive and relatively unstable location. Exposure of the exact mechanisms of an alkene-generating reagent with the moniker of gem-dizinc may resolve this problem, report Shinsuke Komagawa and Masanobu Uchiyama from the RIKEN Advanced Science Institute in Wako and their colleagues from Kyoto University and The University of Tokyo.
To transform terminal carbon–oxygen double bonds, or carbonyl groups, into alkenes via a short-lived complex that connects two carbon atoms together, chemists classically turn to organophosphorus salts known as Wittig reagents. Sometimes, however, these reagents fail to react with carbonyls or give unwanted by-products, fueling a search for new substances with improved activity and better structural control.
gem-Dizinc compounds are Wittig-type reagents that can produce a wide range of terminal alkenes quickly and at room temperature. They consist of twin zinc atoms that sandwich a methylene (CH2) unit. Despite these synthetic advantages, the use of gem-dizinc reagents remains limited because their structures are unstable and their modes of action controversial; some mechanistic features have eluded discovery for nearly forty years.
Komagawa, Uchiyama, and their team tackled this challenge by first using detailed spectroscopic experiments to identify the active form of the metal complex. Dizinc compounds can readily react with each other to make dimers, polymers, or cyclic structures. However, their measurements conclusively demonstrated that the single monomer was the dominant chemical species.
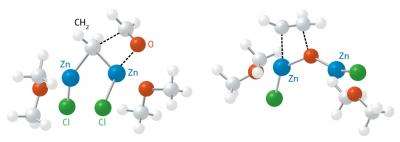
The researchers took this information as the starting point for sophisticated density functional theory calculations of the reaction pathways. Their simulations showed that alkene formation takes place in two steps (Fig. 1): initially, gem-dizinc adds to the carbonyl and forms a cyclic complex. Then, the carbon–carbon double bond is created after gem-dizinc swaps its methylene unit for an oxygen atom. They found that the key factor in making this process so efficient was a cooperative ‘push–pull synergy’ between zinc metals that drove the transformation without having to shuffle electrons between different atoms, making this process quicker than other approaches.
According to Komagawa, these results should help spur the logical design of even better complexes. “The comprehensive mechanistic knowledge acquired in this approach will drive the next stage of this chemistry—more efficient metal reagents that improve the yield and selectivity of alkene formation,” he says.
More information: Sada, M., Komagawa, S., Uchiyama, M., Kobata, M., Mizuno, T., Utimoto, K., Oshima, K. & Matsubara, S. Reaction pathway of methylenation of carbonyl compounds with bis(iodozincio)methane. Journal of the American Chemical Society 132, 17452–17458 (2010). pubs.acs.org/doi/abs/10.1021/ja104439w
Provided by RIKEN