December 16, 2009 weblog
New way to break some of the strongest chemical bonds
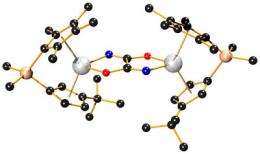
(PhysOrg.com) -- Scientists at Cornell University in the U.S. have found a new way of breaking two of the strongest chemical bonds, at ambient temperature and pressure, and this breakthrough could lead to low-energy processes for making organic compounds containing nitrogen.
Professor of Chemistry at Cornell, Paul Chirik, has developed a new way of making nitrogen-carbon bonds using molecular nitrogen and carbon monoxide, a reaction that would normally need a great deal of energy. N-C bonds are usually created using ammonia as the nitrogen source, but this reaction also needs a large amount of energy. Professor Chirik said the bond between nitrogen and carbon is found in most popular pharmaceuticals, in fertilizers, insecticides, nylon, and in every protein.
Industrial processes use ammonia because the triple bond in molecular nitrogen is too difficult to break. Ammonia is made through the Haber-Bosch process, which takes place at high temperatures and pressure, and uses hydrogen usually sourced from fossil fuels.
Molecular nitrogen (dinitrogen) consists of two nitrogen atoms bound by a triple bond, and is one of the most stable molecules in existence because it has no positive or negative end and the bond between the atoms is extremely strong and difficult to break, and Chirik said "it's very hard to make it react." Carbon monoxide is also extremely stable with strong bonds, and would not react directly with nitrogen.
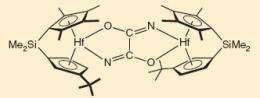
In nature, molecular nitrogen is "fixed" by biological enzymes, and other groups of chemists are working on mimicking the process to produce ammonia that could then be used to provide the nitrogen needed for organic compounds. Chirik's reaction bypasses the ammonia and makes organic compounds containing nitrogen directly.
The reaction takes place in two steps. The first step uses a metal complex (hafnocene), based on hafnium, to react with the nitrogen molecule, breaking two of the three bonds and forming an intermediate chemical consisting of N2 complexed between two hafnocenes. In the second step carbon monoxide is added and reacts with the intermediate compound, breaking the third N-N bond and forming new C-N and C-C bonds. The result is an organic molecule, oxamide (C2H4N2O2), which is released from the hafnium complex when acid is added. Oxamide is an important slow-release fertilizer. If the amount of carbon monoxide is varied, different compounds could be formed instead of oxamide.
The Cornell reaction is not catalytic, one hafnocene complex is used up for every N2 molecule cleaved, and the amounts of oxamide created are not large enough to be useful, so as it stands the reaction is impractical for industrial use. Chirik said they are working on "how to get the pieces off the metal" to allow the hafnocene to be reused. They are also trying to work out if the same kind of reaction could be used to produce other nitrogen-organic compounds, and if complexes using metals other than the rare hafnium could be used.
If the Cornell process becomes viable for industrial applications it could be in great demand, since nitrogen is so abundant, making up 78 percent of the atmosphere, and because Chirik's reaction takes place at room temperature and pressure.
Chirik's paper is published in the December 13 issue of the journal Nature Chemistry.
• Join PhysOrg.com on Facebook!
• Follow PhysOrg.com on Twitter!
More information: D J Knobloch, E Lobkovsky and P J Chirik, Nature Chemistry, 2009, DOI:10.1038/nchem.477
© 2009 PhysOrg.com
















