Gasoline from water, CO2 and sunlight

A research team from ETH Zurich, PSI, and Caltech demonstrates the stable, efficient and rapid generation of solar fuels. They apply a thermochemical cycle based on ceria redox reactions to thermochemically split water and CO2 using a novel solar cavity-receiver reactor under concentrated solar radiation. Their remarkable findings are reported in the latest issue of Science.
Scientists ask themselves: how can we get hold of the vast, yet intermittent and unevenly distributed, solar energy resource such that it can be stored and transported from the sunny and uninhabited regions of the earth’s sunbelt to the world’s industrialized and populated centers, where much of the energy is required? This question has motivated the search for recipes to transform sunlight into chemical energy carriers in the form of storable and dispatchable liquid fuels, such as gasoline and jet fuel, usable to propel not only our cars, airplanes and ships, but the entire world economy.
New recipe and cooking pot
A research team around Aldo Steinfeld, Professor of Mechanical and Process Engineering at ETH Zurich and Head of the Solar Technology Laboratory at PSI, in collaboration with the California Institute of Technology (Caltech) in Pasadena, USA, has recently developed a promising recipe and associated reactor technology. Their idea is based on a solar-driven thermochemical cycle for splitting CO2 and H2O using metal oxide redox reactions. The 2-step cycle consists in thermally reducing non-stoichiometric cerium oxide at above 1,500 °C and re-oxidizing it with H2O and CO2 at below 900 °C to produce H2 and CO – known as syngas, the precursor of liquid hydrocarbon fuels. “The operation at high temperatures and the utilization of the entire solar spectrum provide a thermodynamically attractive path to solar fuel production at high kinetic rates and energy conversion efficiencies”, says Steinfeld.
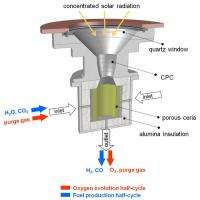
The solar reactor for performing this cycle is schematically shown in the adjacent figure. It consists of a cavity-receiver with a small windowed aperture for the access of concentrated solar radiation. The selected dimensions ensure multiple internal reflections and efficient capture of incoming solar energy. A porous, monolithic ceria cylinder is placed inside the cavity and subjected to multiple heat-cool cycles under appropriate gases to induce fuel production.
Efficient heat transfer
With this arrangement, the porous ceria cylinder is directly exposed to concentrated solar radiation impinging on its inner walls, providing efficient radiative heat transfer directly to the reaction site. Reacting gases flow radially across the porous ceria cylinder, while product gases exit the cavity through an axial outlet port. Experimentation was carried out at PSI’s High-Flux Solar Simulator with a 2000 W solar reactor prototype subjected to solar concentration ratios exceeding 1,500 suns. The measured solar-to-fuel energy conversion efficiency, defined as the heating value of the fuel produced divided by the solar radiative power input, reached 0.8 %.
“This efficiency value is about two orders of magnitude greater than the one observed with state-of-the-art photocatalytic approaches for CO2 dissociation”, says Philipp Furler, doctoral student in Steinfeld’s group, who is currently working on the reactor optimization with help of fluid mechanics and heat transfer simulation models. Thermodynamic analyses show that efficiencies above 15% are attainable.
500 cycles without interruption
Beyond efficiency, material stability is an crucial criterion for a viable thermochemical process. Using the differential reactor system, 500 cycles of water dissociation were performed without interruption, yielding stable fuel production at constant rates.
Essential to the successful solar fuel demonstration is a simple and scalable solar reactor design using porous ceria that enables high-temperature heat transfer to the reaction sites under realistic operating conditions relevant to large-scale solar concentrating systems. In the meantime, Steinfeld and his team are currently focusing on optimizing the solar reactor technology with the aim of scaling it up for megawatt solar towers, such as those already applied commercially for electricity generation.
When asked about the timetable towards industrial implementation, he is cautious and rather conservative: “The potential of solar fuels has been repeatedly overestimated in the short term and underestimated in the long term. We still have plenty of R&D work to accomplish, but by 2020 we should be able to witness the first industrial solar fuel plants coming into operation”. He confidently adds: “The results to date provide compelling evidence for the technical viability of thermochemical approaches to solar fuel generation from water and CO2, while clarifying the efforts required to transform the laboratory concept into a central technology for a sustainable energy future”.
More information: Chueh WC, et al. High-Flux Solar-Driven Thermochemical Dissociation of CO2 and H2O using Nonstoichiometric Ceria. Science, 24 December 2010: Vol. 330 no. 6012 pp. 1797-1801. DOI:10.1126/science.1197834
Provided by ETH Zurich















