New catalyst of platinum nanoparticles could lead to conk-out free, stable fuel cells
In the quest for efficient, cost-effective and commercially viable fuel cells, scientists at Cornell University's Energy Materials Center have discovered a catalyst and catalyst-support combination that could make fuel cells more stable, conk-out free, inexpensive and more resistant to carbon monoxide poisoning.
The research, "Highly Stable and CO-Tolerant Pt/Ti0.7W0.3O2 Electrocatalyst for Proton-Exchange Membrane Fuel Cells," (Journal of the American Chemical Society, July 12, 2010) led by Hector D. Abruna, Cornell professor of Chemistry and Chemical Biology and director of the Energy Materials Center at Cornell (emc2); Francis J. DiSalvo, Cornell professor Chemistry and Chemical Biology; Deli Wang, post doctoral researcher; Chinmayee V. Subban, graduate student; Hongsen Wang, research associate; and Eric Rus, graduate student.
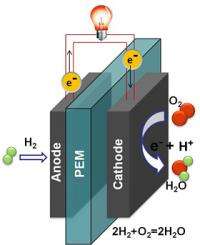
Hydrogen fuel cells offer an appealing alternative to gasoline-burning cars: They have the potential to power vehicles for long distances using hydrogen as fuel, mitigate carbon dioxide production and emit only water vapor.
However, fuel cells generally require very pure hydrogen to work. That means that conventional fuels must be stripped of carbon monoxide - a process that is too expensive to make fuel cells commercially viable.
Fuel cells work by electrochemically decomposing fuel instead of burning it, converting energy directly into electricity.
The problem is that platinum and platinum/ruthenium alloys, which are often used as catalysts in PEM (proton exchange membrane) fuel cells, are expensive and easily rendered ineffective by exposure to even low levels of carbon monoxide.
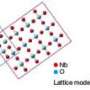
To create a catalyst system that can tolerate more carbon monoxide, Abruna, DiSalvo and colleagues deposited platinum nanoparticles on a support material of titanium oxide with added tungsten to increase its electrical conductivity.
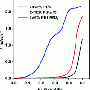
Their research shows that the new material works with fuel that contains as much as 2 percent carbon monoxide - a level that is about 2000 times that which typically poisons pure platinum. Also, the material is more stable and less expensive than pure platinum. With the new catalyst, said Abruña, "you can use much less-clean hydrogen, and that's more cost-effective because hydrogen derived from petroleum has a very high content of carbon monoxide. You need to scrub off the carbon monoxide and it's very expensive to do that."
The researchers are now preparing to put the catalyst to the test in real fuel cells. "So far, indications are very good," Abruña said.
In preliminary experiments comparing the new material's performance with pure platinum, he added, the platinum cell was readily poisoned by carbon monoxide and conked out early. Said Abruña: "But ours was still running like a champ."
Provided by Cornell University