Where droplets gently touch a membrane
(PhysOrg.com) -- Little or nothing happens in cells without the involvement of the minute membrane vesicles: for example, vesicles act as recycling centres for cell waste, as detoxification stations and as a vehicle for substance transport.
Scientists at the Max Planck Institute of Colloids and Interfaces have now described mathematically why some vesicles constrict to form a figure-eight shape. As part of this process, they have established that this constriction differs in its details to what was previously thought, and that this shape is dependent on the material characteristics of the components involved. These types of constricted vesicles may also play a role in vesicle formation in biological cells, as well as in the separation of cytoplasm into compartments with different protein contents. (Physical Review Letters, December 4, 2009)
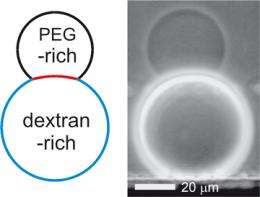
Membrane vesicles are usually spherical in form. However, they sometimes arise as double vesicles with the outline of a figure-eight - for example, when one vesicle separates off from another. "Viewed under an optical microscope, the two vesicles appear to be separated by a sharp incision," says Halim Kusumaatmaja, who played a major role in the current study at the Max Planck Institute of Colloids and Interfaces in Potsdam-Golm. Researchers would expect to find this in the case of two droplets in contact without a membrane. However, when a membrane surrounds the droplets, the constriction should curve smoothly. The fact that it does precisely this at nanoscopic level is what the Potsdam researchers have succeeded in demonstrating with the help of theoretical and empirical tests.
The extent to which the constriction and the phase boundary curve depends on the components involved: the substances dissolved in the droplets, the solvent and the characteristics of the membrane. This is where things become rather complicated, and without first understanding how the double-bellied vesicles are formed, it is difficult to progress any further: the Potsdam researchers studied vesicles containing a mixed solution of two different polymers. Both polymers can be dissolved easily in water, but they try to stay out of each other’s way. The vesicles, in turn, float in another solution that is as strong as the solution found inside them. This is how things would remain if the researchers did not add an additional component to the exterior solution, which generates an imbalance in osmotic pressure: the exterior solution then contains more particles per litre than the solution inside the vesicles. As a result, water exits the vesicles through the membranes and enters the exterior solution.
Due to the water loss, the solution inside the vesicle becomes more concentrated - so strong that the two polymers, which are not particularly compatible, move closer to each other than they would have liked. As a result, the fluid in the vesicle divides into one part that contains more of one polymer and another with a higher proportion of the second polymer. Physicists refer to this phenomenon as phase separation. "It is possible that in cells different compartments are also formed in this way," says Halim Kusumaatmaja. In any case, the two fluids or phases in the vesicle are now separated from each other and from the fluid outside the vesicle.
The different chemical compositions of the three solutions also give rise to a different physical behaviour - in this particular case, different surface tensions at the boundaries. In a similar way in which that a drop of water assumes a more spherical form on a hydrophobic surface than it does on a hydrophilic one, on which it may spread completely in extreme cases, the surface tensions between the two fluids and the membrane, which is surrounded by a different fluid, determine the curvature at the vesicle borders. An important distinction in the latter case is that the stiffness of the membrane also plays a role.
By examining the balance of forces involved, the scientists then developed a new concept of an intrinsic contact angle, which arises from the characteristics of the fluids and the membrane. This quantity, however, is not measurable directly and hence it represents a ‘hidden variable’ within the system. "We were able however to establish a link between this contact angle and some measurable parameters," says Kusumaatmaja. Careful comparisons with experiments provided evidence that the intrinsic contact angle is indeed a genuine and useful material parameter.
"This system is of particular interest to us as it enables us to observe the interplay between wetting processes and membrane physics," says Reinhard Lipowsky who, as a Director of the Potsdam-based Institute, heads the research. Wetting processes arise when a fluid comes into contact simultaneously with a gas or another fluid and a solid interface.
Circumstances change, of course, when the wetting of a flexible membrane is involved rather than that of a solid membrane. Even with two connected droplets with different contents floating in a third fluid without an enclosing membrane, the constricted membrane vesicles are not comparable: "In this kind of system, a sharp kink would actually arise between the droplets," says Halim Kusumaatmaja. "The gently curved constriction only arises with the membrane."
Based on the current findings, the scientists would now like to further study how the shape of the vesicles depends on the intrinsic contact angle and the tension between the two solutions inside the vesicle. "We would like to fully understand the behaviour of such systems in order to learn even more about vesicle formation in biological cells", says Reinhard Lipowsky. This information could also facilitate, for example, the handling of fluids on microchips in laboratories. Such minute analytical devices enable the implementation of sensitive and simple analyses in chemistry, biology and medicine. It may be possible to separate a fluid in this way into droplets with different contents, as the Potsdam-based researchers succeeded in doing in their basic investigations.
More information: Halim Kusumaatmaja, Yanhong Li, Rumiana Dimova, and Reinhard Lipowsky, Intrinsic Contact Angle of Aqueous Phases at Membranes and Vesicles, Physical Review Letters, December 4, 2009
Provided by Max-Planck-Gesellschaft