Opposing functions of a key molecule in the development of organisms
Scientists headed by ICREA researcher Marco Milán, at the Institute for Research in Biomedicine (IRB Barcelona, Spain), reveal a surprising new function of Notch protein that contrasts with the one known to date. Found in the cell membrane, this protein activates a signalling pathway that regulates the expression of genes that make the cell divide, grow, migrate, specialise or die. Notch activity is required for the correct development of organisms and for the maintenance of tissues in adults. When Notch acts at an incorrect time or in an incorrect context, it can give rise to the generation of tumours, among these leukaemia, breast cancer, colon cancer, skin cancer, lung cancer and renal carcinomas.
"The same pathways responsible for the development and growth of organisms are involved in the transformation of healthy cells into cancerous ones", says Marco Milán, so "all new data on the modulation of Notch activity, the first step in the chain, may be relevant for the design of effective therapies". Marco Milán's group has now discovered that the presence of Notch proteins in the cell membrane is also required to inactivate the pathway. The description of the new role of Notch, found in the fly Drosophila melanogaster, and the mechanism that regulates this function have been published in the journal Current Biology, which belongs to the Cell group.
Stop! Notch is a double agent
In order for the Notch pathway to be activated, ligand-type proteins from neighbouring cells bind to the Notch receptor. When the ligand and receptor come into contact, the Notch receptor is processed and the intracellular part moves to the nucleus to activate gene expression. This is the basic and "extremely simple activation system" of the Notch signalling pathway, which is based on short distance contact between cells through a ligand and a receptor.
In a developing wing and through a technique called Clonal Analysis, the researchers manipulated groups of cells, among groups of normal cells, to remove Notch receptor expression. The scientists used the Drosophila wing because it is an excellent model to describe how cells behave when a certain gene is mutated and to determine and test how this mutation affects adjacent cells. This was the objective of the study designed by Isabelle Becam, post-doctoral researcher in Milán's Group and first author of the article. "As expected, the cells lacking Notch did not activate the pathway, but what was surprising was the observation that neighbouring cells did". Becam then questioned whether the absence of Notch in a group of mutated cells could cause activation.
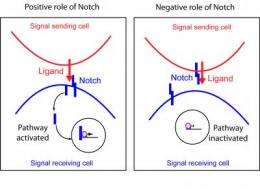
Indeed, the analyses demonstrated that the Notch receptor sequesters the ligands and prevents these from connecting to the Notch receptors of adjoining cells. The experiments showed that the absence of the receptor in the mutated cells leaves many ligands free, ready to enter into contact with Notch receptors of the non-manipulated cells. "It is strange, but in the cell emitting the signal, Notch receptor captures the ligands by acting as a silencer while in the cell receiving the signal the binding of ligands with Notch allows activation of the pathway". "In fact", says Milán, "it is all to do with a fine balance between ligands and receptors of the emitting and receiving cell". In other words, Notch is a kind of double agent and exerts opposing functions: repressing or activating the pathway depending on whether it is located in cells emitting or receiving the signal. It must be noted that such a simple activation system involves multiple repression mechanisms, "because this is a crucial but also dangerous signalling pathway", explains Milán.
The researchers have discovered the self-repression mechanism of Notch in Drosophila and it should be checked now whether this also operates in mice and humans. They speculate that it does because the ligand-receptor system of Notch activation has been conserved in all organisms. "If this new mechanism is also present in vertebrates, it should be taken into consideration when designing effective therapies against certain kinds of cancer, such as T-cell acute lymphoblastic leukaemia (T-ALL)", concludes Milán.
It is well established that the Notch pathway controls the development of T lymphocytes, cells of the immune response system found in blood. The cells destined to become lymphocytes receive the appropriate signalling through Notch receptors. In more than half T-ALL patients the Notch receptor is permanently activated in the T-cell precursors. Thus the continuous proliferation of cells is stimulated until tumours form. "A priori, blocking the Notch receptor could appear to be a good strategy to combat this kind of leukaemia. However, the results of our work suggest that blocking the receptor only in some cells would cause undesirable effects in adjacent cells", warns Milán.
More information: A role of Notch in ligand cis-inhibition in Drosophila. Isabelle Becam, Ulla-Maj Fiuzza, Alfonso Martínez-Arias and Marco Milán. Current Biology, 2009. doi: 10.1016/j.cub.2010.01.058
Provided by Institute for Research in Biomedicine (IRB Barcelona)















