Ocean waves may hold secret to efficient renewable energy

As the demand for renewable wind and solar energy steadily increases, the need to reduce the cost and extend the life of renewable energy storage batteries becomes even greater.
By getting back to the basics, a University of Cincinnati quantum chemistry researcher looks at how water and other molecules align and influence ionic distribution on the surface where air and the liquid meet. These findings have received respect from top physicists around the globe and show promise for enhancing the efficiency of renewable energy devices.
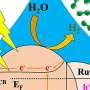
As part of his National Science Foundation-funded research titled "Computational Studies of Specific Ion effects in Water and Ion Channels," Thomas Beck, professor of chemistry in UC's McMicken College of Arts and Sciences, leveraged the technical resources of the Ohio Supercomputer Center (OSC) to improve methods for calculating the thermodynamics of ion hydration. In other words, to better understand the general concepts inside batteries and renewable energy storage systems—for energy generated from sources such as concentrated solar and photovoltaic solar—Beck found a much more efficient way to quantify the electrical voltage at the surface of water and other liquids like ethylene carbonate and propylene carbonate.
Beck has developed basic methods using quantum calculations to show that older, more traditional methods for gauging water's electrical surface charge often over-estimate the polarization state of ions, which is at the center of the electrical processes.
"Scientists have historically tried to simulate ions in water by developing force fields for use in molecular mechanics, but they associate those findings with experimental data that may or may not include the electrical potential at the water's surface," says Beck. "These models have often led to imprecise measurements and distorted values.
"By understanding what is actually happening at a molecular level on the liquid's surface, we can improve the general concepts for interfaces inside renewable energy devices."
Ultimately, Beck feels that progress is being made in wind and solar renewable energy but may not be fast enough for the crisis that is expected. Given the trajectory that energy is being used at present rates, he predicts that future energy demands may likely double in the next 50 years, in part because of the growth of developing countries.
"Up to two-thirds of the available fossil fuel sources of the Earth's energy will likely be depleted on a time scale of 50 to 100 years," says Beck. "This underscores the need for changing our energy infrastructure and why we are working diligently to develop better future energy resources."
Ocean tide's role in energy
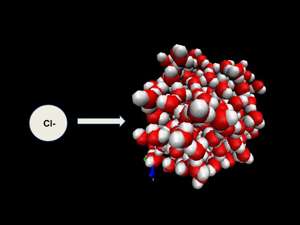
As an example of the powerful role hydration ions play in energy, Beck points to the atmosphere above an ocean. He explains that every time a wave breaks, droplets of water containing ions—molecules that have either a positive or negative charge—go up in the air, but how those ions arrange near the surface of the droplets can affect the chemistry of what happens in the atmosphere around them. Those ions can affect when light comes in and how it is absorbed or reflected, all of which can affect the atmospheric temperature. So hidden in this very general atmospheric chemistry can lie some of the most profound answers for energy.
Beck's enhanced methods for gauging the thermodynamics of ion hydration have garnered an overwhelming international respect from many of the world's foremost physicists who have invited him to speak at several prestigious quantum chemistry and physical society conferences around the globe, from Norway to Australia and now Switzerland.
This September, Beck has been invited to give the keynote address in Lausanne, Switzerland, to a cutting-edge experimental workshop group at the Surface Potentials - Facts, Findings and Fantasies workshop. And he will present on the surface potential of water titled, "The Role of the Water Surface Potential in Ion Specificity." This prestigious group includes two theoreticians and several of the top experimental scientists in the world, all of whom are trying to quantify or characterize this electrical-surface potential.
Beck's contention is that the electrical potential right near the surface of water actually affects the chemistry of the surrounding interior water. And the fundamental mechanism behind the chemical change at the surface is crucial and needs to be further understood.
"The relevance of this topic to the Swiss conference will add to the big debate about whether the surface of water carries a positive or a negative charge and will also lead in to our investigation of how ions distribute at the surface of fresh versus salt water."
Beck also has developed a practical way of applying this fundamental ionic process to how ions are arranged inside batteries near the interface, which ultimately affects how the battery works.
"In my research I show a plot as a function of time where the estimates for that electrical surface potential jump around a lot," explains Beck. "By the very nature of the problem it becomes very complicated, but essentially, when I take a hypothetical charge that doesn't interact with anything and I drag it across the water's surface, it turns out that the voltage change is about four volts (-0.4), which is a huge potential increase from the one to two volts (-0.1 to -0.2) others have estimated before.
"This was a shocker that was only discovered in the last five years."
Beck is also currently involved in related research investigating the thermodynamics of ion solvation in the non-aqueous solvents ethylene carbonate and propylene carbonate, both of which are commonly used in renewable energy super capacitors.
Provided by University of Cincinnati