March 3, 2015 feature
Na-ion batteries get closer to replacing Li-ion batteries
(Phys.org)—As lithium resources continue to decline worldwide, the next generation of portable electronics will most likely be powered by something other than Li-ion batteries. One potential candidate is the sodium-ion (Na-ion) battery, which stands out because sodium is cheaper, non-toxic, and more abundant than lithium.
Currently, one of Na-ion's largest drawbacks is that the batteries take a long time to charge and discharge, and a slow discharge rate does not supply enough power density for high-power applications. In general, there is a tradeoff between the charge/discharge rate and capacity, so that attempts to increase the charge/discharge rate have resulted in severely reduced capacity.
Now in a new study published in the Journal of the American Chemical Society, researchers led by Yong Lei, a professor at the Technical University of Ilmenau in Germany, have achieved a significant improvement in this area. The researchers demonstrated a Na-ion battery that exhibits charge/discharge rate and capacity values that are among the highest achieved for both organic Na-ion and Li-ion batteries. The large improvement may help pave the way toward the integration of Na-ion batteries in portable and wearable electronics.
To achieve this improvement, the researchers had to consider some of the fundamental properties of sodium. Sodium and lithium have a similar tendency to lose electrons—as measured by their electrochemical potential—which makes them good anode materials. However, sodium ions are nearly 25% larger than lithium ions. The larger size makes it more difficult for sodium ions to be inserted into the crystal structure of the electrodes, where the chemical reactions take place. As a result, the ions can't move as fast, which lies at the root of the slow charge/discharge problem. Tied in with this problem, the charge transport and stability of the materials also need to be improved.
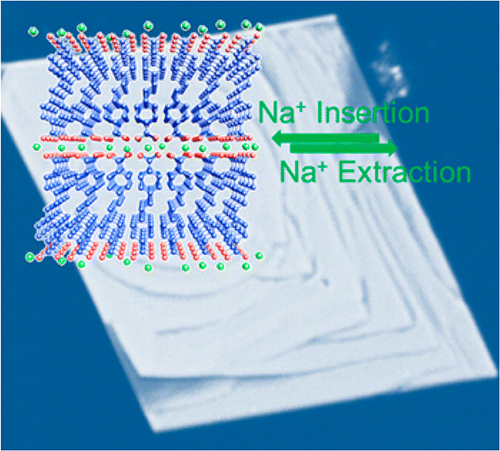
Although the researchers could not decrease the size of the sodium ions, they could improve the efficiency with which the sodium ions are inserted into the electrodes. To do this, the researchers used a molecular design strategy based on extending the electrode material's π-conjugated system, which basically involves manipulating the way that these molecules bond with each other. Physically, this strategy results in a terrace morphology, consisting of multiple, widely spaced layers that form a faster route for the sodium ions to move through. The extended π-conjugated system also improves the charge transport and stabilizes the charged/discharged states so that they can better tolerate the fast insertion/extraction of Na ions.
In terms of battery performance, this change results in significant improvements. As always, there is still a tradeoff between charge/discharge rates and capacity. But the new Na-ion batteries can operate at a current density (a measure of the charge/discharge rate) that is 1000 times higher (10 A/g vs. 10 mA/g) than most previously reported organic Na-ion batteries while retaining a much higher capacity (72 mAh/g).
At an intermediate current density (1 A/g), the new battery delivers an impressive reversible capacity of 160 mAh/g, which is one of the highest values reported for both organic Na-ion and Li-ion batteries to date. The battery also exhibits good capacity retention (70% retention after 400 cycles).
"In this work, we focused on the molecular design for improving battery performance and addressing the current challenge of fast-charge and -discharge in organic Na-ion batteries," Lei told Phys.org. "Through reasonable molecular design strategy, we demonstrated that the extension of the π-conjugated system is an efficient way to improve the high rate performance, leading to much enhanced capacity and cyclability. We also think our work provides a good attempt to expand the search of new electrode materials from the traditional inorganic to organic materials, and might arouse further attention regarding this area."
Building on this research, the scientists plan to further improve the batteries using the molecular design strategy and other innovative methods.
"We will be continuously focusing on organic Na-ion batteries," Lei said. "As mentioned in our paper, there are only a few reports on organic Na-ion batteries. Therefore, there is still a long way to go for organic Na-ion batteries. Future goals include: 1) improve the capacity and cyclability of the cathodes and anodes; 2) enhance the rate performance of the cathodes and anodes; 3) adjust the electrochemical potential to achieve suitable output voltage of full cells; and 4) develop environmentally friendly and low-cost materials."
More information: Chengliang Wang, et al. "Extended π-Conjugated System for Fast-Charge and -Discharge Sodium-Ion Batteries." Journal of the American Chemical Society. DOI: 10.1021/jacs.5b00336
Journal information: Journal of the American Chemical Society
© 2015 Phys.org