Regulation of developmental gene expression occurs in the reverse order to that expected

Molecular modification of DNA and the histone proteins it is bound to is one of the key mechanisms responsible for regulating gene expression. Polycomb-group proteins play an important role in some of these modification processes, but the molecular mechanisms by which they bind to DNA and exert their effects have been poorly understood.
Takashi Kondo and colleagues from the Laboratory of Developmental Genetics at the RIKEN Center for Integrative Medical Sciences, in collaboration with researchers from the University of Oxford in the United Kingdom, have now shown that modifications involving Polycomb proteins occur in a manner that contradicts existing models.
Polycomb-group proteins are found in two complexes, known as PRC1 and PRC2, and there are two forms of PRC1—the canonical and variant forms. Molecular modifications involving these complexes play crucial roles in gene regulation, cellular differentiation and development. "Polycomb-group proteins repress gene transcription by binding to DNA," says Kondo. "This is the main system that regulates transcription of developmental genes, and Polycomb systems are also related to some cancers. Understanding their mechanism of action is therefore biologically very significant."
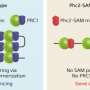
PRC1 and PRC2 complexes always work together, but exactly how has remained unclear. For some time, it has been thought that PRC2 must first bind to the DNA, which then allows binding or 'recruitment' of PRC1. However, as recent evidence suggests that this might not be the case, Kondo and his colleagues developed a new approach to investigate.
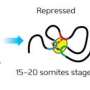
The researchers inserted human DNA into mouse embryonic stem cells to cause the PRC1 complex to bind at specific sites. They found that the variant form of PRC1, but not the canonical form, was able to recruit PRC2 to the DNA—the reverse of the long-accepted mechanism of PRC2 binding prior to recruiting PRC1.
This process is dependent on a component of the PRC1 complex called KDM2B, and deleting part of this protein prevented PRC1 from binding to DNA in cells. In live mice, genetically disrupting the function of KDM2B, thereby preventing PRC1 binding and PRC2 recruitment, had serious consequences: complete loss of KDM2B function was lethal before birth, while partial loss caused abnormalities in the skeleton due to incorrect development.
"Our findings indicate that the Polycomb regulatory mechanisms possibly rely on the activity of variant PRC1 rather than PRC2 or canonical PRC1," explains Kondo. "This means that studies on variant PRC1 may be more fruitful for investigating the mechanisms of developmental regulation and cancer development."
More information: Blackledge, N. P., Farcas, A. M., Kondo, T., King, H. W., McGouran, J. F., Hanssen, L. L. P., Ito, S., Cooper, S., Kondo, K., Koseki, Y. et al. "Variant PRC1 complex-dependent H2A ubiquitylation drives PRC2 recruitment and polycomb domain formation." Cell 157, 1445–1459 (2014). DOI: 10.1016/j.cell.2014.05.004
Journal information: Cell
Provided by RIKEN