Scientists take steps to unlock the secrets to the fountain of youth
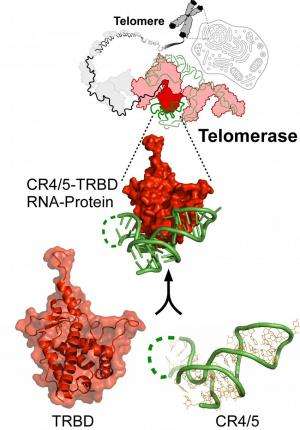
ASU scientists, together with collaborators from the Chinese Academy of Sciences in Shanghai, have published today, in Nature Structural and Molecular Biology, a first of its kind atomic level look at the enzyme telomerase that may unlock the secrets to the fountain of youth.
Telomeres and the enzyme telomerase have been in the medical news a lot recently due to their connection with aging and cancer. Telomeres are found at the ends of our chromosomes and are stretches of DNA which protect our genetic data, make it possible for cells to divide, and hold some secrets as to how we age –and also how we get cancer.
An analogy can be drawn between telomeres at the end of chromosomes and the plastic tips on shoelaces: the telomeres keep chromosome ends from fraying and sticking to each other, which would destroy or scramble our genetic information.
Each time one of our cells divides its telomeres get shorter. When they get too short, the cell can no longer divide and it becomes inactive or dies. This shortening process is associated with aging, cancer and a higher risk of death. The initial telomere lengths may differ between individuals. Clearly, size matters!
"Telomerase is crucial for telomere maintenance and genome integrity," explains Julian Chen, professor of chemistry and biochemistry at ASU and one of the project's senior authors. "Mutations that disrupt telomerase function have been linked to numerous human diseases that arise from telomere shortening and genome instability."

Chen continues that, "Despite the strong medical applications, the mechanism for telomerase holoenzyme (the most important unit of the telomerase complex) assembly remains poorly understood. We are particularly excited about this research because it provides, for the first time, an atomic level description of the protein-RNA interaction in the vertebrate telomerase complex."
More information: Structural basis for protein-RNA recognition in telomerase, DOI: 10.1038/nsmb.2819
Journal information: Nature Structural and Molecular Biology
Provided by Arizona State University


















