Cationic small molecules hold great potential for preventing and treating fungal infections
Pathogenic fungi like Candida albicans can cause oral, skin, nail and genital infections. While exposure to pathogenic fungi is generally not life-threatening, it can be deadly to immunocompromised patients with AIDS or cancer. A variety of antifungal medications, such as triazoles and polyenes, are currently used for treating fungal infections. The range of these antifungal medications, however, is extremely limited, with some fungal species developing resistance to these drugs.
Yi Yan Yang at the A*STAR Institute of Bioengineering and Nanotechnology in Singapore and co-workers, in collaboration with IBM Almaden Research Center in the United States, have discovered four cationic terephthalamide-bisurea compounds with strong antifungal activity, excellent microbial selectivity and low host toxicity. These small molecules can self-assemble into fibers, bind fungal membrane and rupture the cells of a variety of fungal species. The results may expand the possibilities of medication for battling drug-resistant fungal species.
Conformational analysis revealed that the terephthalamide-bisurea compounds have a Z-shaped structure: the terephthalamide sits in the middle, urea groups on both sides of the terephthalamide, and cationic charges at both ends. The researchers prepared compounds with different spacers—ethyl, butyl, hexyl or benzyl amine—in-between the urea group and the cationic charge.
When dissolved in water, the terephthalamide-bisurea compounds aggregate to form fibers with lengths ranging from a few hundred nanometers to several micrometers. Some of the compounds form fibers with high flexibility and others with high rigidity.
The researchers evaluated the antifungal activity of their terephthalamide-bisurea compounds against C. albicans. They found that all of the cationic compounds effectively inhibited fungal growth, even when the fungal concentration increased from 102 to 105 colony-forming units per milliliter.
The researchers believe that the potent antifungal activity is largely due to the formation of fibers with extremely small diameters in the order of 5 to 10 nanometers, which facilitates the rupture of fungal membranes. "This is particularly important because the fungal membrane of C. albicans is multilayered and has low negative charges," explains Yang. "It also helps explain why cationic terephthalamide-bisurea compounds could easily penetrate the fungal membrane."
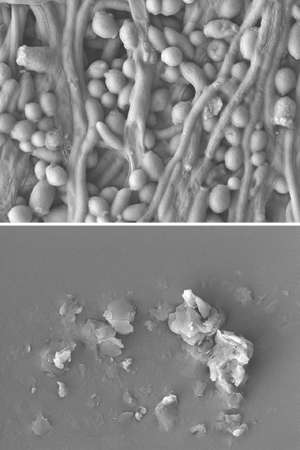
The terephthalamide-bisurea compounds also eradicated clinically isolated drug-resistant C. albicans. The compounds prevent the development of drug resistance by rupturing the fungal membrane of C. albicans and disrupting the biofilm (see image).
Additionally, cytotoxicity tests showed that the cationic terephthalamide-bisurea compounds exhibit low toxicity toward mammalian cells and in a mouse model, revealing that the compounds "are relatively safe for preventing and treating fungal infections," says Yang.
More information: Fukushima, K., Liu, S., Wu, H., Engler, A. C., Coady, D. J. et al. "Supramolecular high-aspect ratio assemblies with strong antifungal activity." Nature Communications 4, 2861 (2013). dx.doi.org/10.1038/ncomms3861
Journal information: Nature Communications



















