Addition of pectin molecules strengthens silk biomaterials
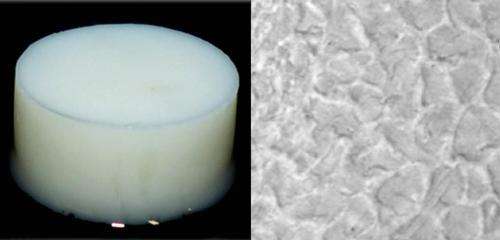
The human body has limited ability to self-repair damage to cartilage or bone. Implantable 'bioscaffold' materials that can be seeded with cells can potentially be used to regenerate these critical tissues. One such biomaterial under consideration is silk hydrogel—a nontoxic, natural substance produced by combining silk proteins with water to form an aqueous gel. Keiji Numata from the RIKEN Center for Sustainable Resource Science and colleagues have now devised a way to reinforce silk-based bioscaffolds through the addition of pectin-based fibers.
Due to their high bound water content, silk hydrogels have been shown to accelerate the growth of human mesenchymal stem cell (hMSC) adhesion proteins. hMSCs can differentiate into bone and cartilage cells and are therefore ideal precursors for tissue regeneration strategies. Unfortunately, silk hydrogels often have insufficient mechanical strength for practical applications.
To boost the elasticity and mechanical strength of silk hydrogels, Numata and his colleagues turned to pectin, a polysaccharide found in plant cells that is widely used as a food thickening agent. The team suspected that silk's arginine and lysine amino acids could interact with pectin to form a strong molecular network (Fig. 1). "Silk is a hydrophobic polymer, while pectin polymers are hydrophilic," says Numata. "The key challenge was to make a stable polymer blend from these two materials."
The researchers obtained an aqueous silk solution by extracting proteins from silkworm cocoons and filtering them through dialysis membranes. They then mixed the silk solution with pectin, sealed the mixture in a container fitted with a dialysis membrane, and immersed the container in methanol. The methanol regulated the displacement of water through the dialysis membrane, producing a semi-solid silk–pectin hydrogel.
Studying the silk–pectin hydrogel under a microscope revealed the formation of a heterogeneous network of silk fibers and random coils of silk proteins associated with the pectin. The hydrogels also displayed greatly improved strength as well as enhanced water-holding capacity, which resulted in the release of water during mechanical compression—an action that mimics natural cartilage behavior. Cell culture experiments showed that hMSCs had exceptional viability on the silk–pectin substrate, and the bioscaffold itself was demonstrated to degrade on exposure to enzymes, encouraging the integration of differentiating cells into surrounding host tissue.
Numata notes that whereas most hydrogels are prepared from purely hydrophilic polymers, the extraordinary strength exhibited by this example of a hydrophobic/hydrophilic mixture suggests that this unconventional strategy could open a new avenue for hydrogel synthesis.
More information: Numata, K., Yamazaki, S., Katashima, T., Chuah, J.-A., Naga, N. & Sakai, T. "Silk-pectin hydrogel with superior mechanical properties, biodegradability, and biocompatibility." Macromolecular Bioscience advance online publication, 7 March 2014 DOI: 10.1002/mabi.201300482
Provided by RIKEN


















