Mixing silicon with other materials improves the diversity of nanoscale electronic devices
The semiconductor silicon lies at the heart of the current revolution in electronics and computing. In particular, it can produce compact integrated circuits when processed by modern techniques capable of fabricating structures just a few nanometers in size.
Now, Man-Fai Ng and Teck Leong Tan at the A*STAR Institute of High Performance Computing in Singapore have shown that mixing silicon with similar materials can open the door to the fabrication of nanoscale devices with a diverse array of properties that have a wider range of applications.
Ng and Tan used state-of-the-art computer simulations to assess the structural stability and electronic properties of silicon-based nanowires. As their name suggests, nanowires are just a few nanometers wide but can be up to a millimeter long. They exhibit unusual electronic properties because their small width confines the motion of electrons across the wire.
The properties of silicon nanowires are well established, but there is considerable scope to expand their applicability. Scientists anticipate they could realize a more diverse range of characteristics by partially replacing silicon with other elements that are in the same column as silicon in the periodic table. There are many potential materials—including carbon, germanium and tin—each of which can be combined with silicon in any ratio to form an alloy.
Consequently, the total number of possible alloys is immense. The researchers thus undertook a comprehensive search of all these silicon-based alloys to determine which are atomically stable and which have the best properties for nanowire devices.
Ng and Tan employed three mathematical techniques (namely, density functional theory, the cluster expansion method and the Monte Carlo method) to simulate different atomic arrangements in nanowires.
"Instead of evaluating all possible alloy structures, our multiscaled simulation approach enabled rapid large-scale comparison of different combinations of alloy structures and selected the thermodynamically stable ones," explained Ng.
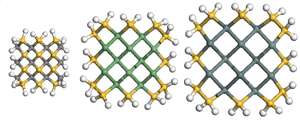
The most stable germanium–silicon and tin–silicon nanowires were found to be those in which the silicon atoms are concentrated around the edge of the wire and the other atomic species are at the core. Conversely, an optimum carbon–silicon nanowire exhibited an ordered arrangement of the atomic species.
Once they had identified the optimum atomic arrangement, Ng and Tan calculated the energy bandgap—a critical parameter for determining the electronic properties of semiconductors. "Next, we plan to improve the bandgap prediction for silicon-based nanowires and develop our approach to address more complicated nanosystems for energy applications," says Ng.
More information: Ng, M.-F. & Tan, T. L. "Unveiling stable group IV alloy nanowires via a comprehensive search and their electronic band characteristics." Nano Letters 13, 4951−4956 (2013). dx.doi.org/10.1021/nl402987c
Journal information: Nano Letters