Research tests which nano system works best cancer treatment

(Phys.org) —In current research related to improving cancer treatments, one promising area of research is the effort to find ways to selectively pinpoint and target cancer cells while minimizing effects on healthy cells.
In that effort, it's already been found in lab experiments that iron-oxide nanoparticles, when heated and then applied specifically to cancer cells, can kill those cells because cancer cells are particularly susceptible to changes in temperature. Increasing the temperature of cancer cells to over 43 degrees Celsius (about 109 degrees Fahrenheit) for a sufficient period of time can kill those cells.
So, a University of Cincinnati-led team – along with researchers at Iowa State University, the University of Michigan and Shanghai Jiao Tong University – recently conducted experiments to see which iron-oxide nanoparticle configurations or arrangements might work best as a tool to deliver this killing heat directly to cancer cells, specifically to breast cancer cells. The results will be presented at the March 3-7 American Physical Society Conference in Denver by UC physics doctoral student Md Ehsan Sadat.
In systematically studying four distinct magnetized nanoparticle systems with different structural and magnetic properties, the research team found that an unconfined nanoparticle system, which used an electromagnetic field to generate heat, was best able to transfer heat absorbed by cancer cells.
So, from the set of nano systems studied, the researchers found that uncoated iron-oxide nanoparticles and iron-oxide nanoparticles coated with polyacrylic acid (PAA) – both of which were unconfined or not embedded in a matrix – heated quickly and to temperatures more than sufficient to kill cancer cells.
Uncoated iron-oxide nanoparticles increased from a room temperature of 22 degrees Celsius to 66 degrees Celsius (about 150 degrees Fahrenheit).
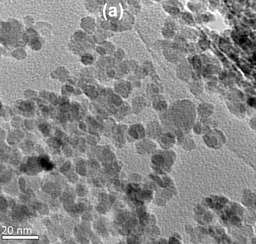
Iron-oxide nanoparticles coated with polyacrylic acid (PAA) heated from a room temperature of 22 degrees Celsius to 73 degrees Celsius (about 163 degrees Fahrenheit.)
The goal was to determine the heating behaviors of different iron-oxide nanoparticles that varied in terms of the materials used in the nanoparticle apparatus as well as particle size, particle geometry, inter-particle spacing, physical confinement and surrounding environment since these are the key factors that strongly influence what's called the Specific Absorption Rate (SAR), or the measured rate at which the human body can absorb energy (in this case heat) when exposed to an electromagnetic field.
According to Sadat, "What we found was that the size of the particles and their anisotropic (directional) properties strongly affected the magnetic heating achieved. In other words, the smaller the particles and the greater their directional uniformity along an axis, the greater the heating that was achieved."
He added the systems' heating behaviors were also influenced by the concentrations of nanoparticles present. The higher the concentration of nanoparticles (the greater the number of nanoparticles and the more densely collected), the lower the SAR or the rate at which the tissue was able to absorb the heat generated.
The four systems studied
The researchers studied
- uncoated iron-oxide nanoparticles
- iron-oxide nanoparticles coated with polyacrylic acid (PAA)
- a polystyrene nanosphere with iron-oxide nanoparticles uniformly embedded in its matrix
- a polystyrene nanosphere with iron-oxide nanoparticles uniformly embedded in its matrix but with a thin film surface of silica
All four nanoparticle systems were exposed to the same magnetic field for 35 minutes, and temperature measurements were performed at two-minute intervals.
As stated, the PAA iron-oxide and the uncoated iron-oxide samples showed the highest temperature change. The lowest temperature changes, insufficient to kill cancer cells, were exhibited by
- The polystyrene nanosphere, which heated to 36 degrees Celsius (about 96 degree Fahrenheit).
- The polystyrene nanosphere with a silica coating heated to 40 degrees Celsius (104 degrees Fahrenheit).
Provided by University of Cincinnati