Clay key to high-temperature supercapacitors
Clay, an abundant and cheap natural material, is a key ingredient in a supercapacitor that can operate at very high temperatures, according to Rice University researchers who have developed such a device.
The Rice group of materials scientist Pulickel Ajayan reported today in Nature's online journal, Scientific Reports that the supercapacitor is reliable at temperatures of up to 200 degrees Celsius (392 degrees Fahrenheit) and possibly beyond. It could be useful for powering devices for use in extreme environments, such as oil drilling, the military and space.
"Our intention is to completely move away from conventional liquid or gel-type electrolytes, which have been limited to low-temperature operation of electrochemical devices," said Arava Leela Mohana Reddy, lead author and a former research scientist at Rice.
"We found that a clay-based membrane electrolyte is a game-changing breakthrough that overcomes one of the key limitations of high-temperature operation of electrochemical energy devices," Reddy said. "By allowing safe operation over a wide range of temperatures without compromising on high energy, power and cycle life, we believe we can dramatically enhance or even eliminate the need for expensive thermal management systems."
A supercapacitor combines the best qualities of capacitors that charge in seconds and discharge energy in a burst and rechargeable batteries that charge slowly but release energy on demand over time. The ideal supercapacitor would charge quickly, store energy and release it as needed.
"Researchers have been trying for years to make energy storage devices like batteries and supercapacitors that work reliably in high-temperature environments, but this has been challenging, given the traditional materials used to build these devices," Ajayan said.
In particular, researchers have struggled to find an electrolyte, which conducts ions between a battery's electrodes, that won't break down when the heat is on. Another issue has been finding a separator that won't shrink at high temperatures and lead to short circuits. (The separator keeps the electrolyte on the anode and cathode sides of a traditional battery apart while allowing ions to pass through).
"Our innovation has been to identify an unconventional electrolyte/separator system that remains stable at high temperatures," Ajayan said.
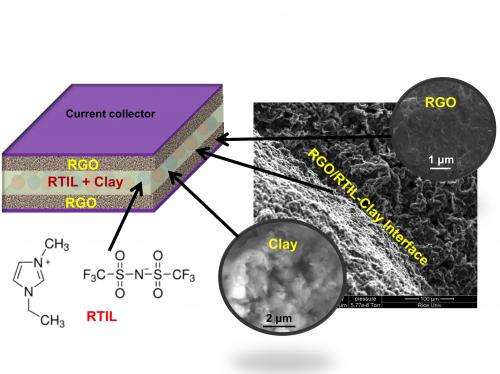
The Rice researchers led by Reddy and Rachel Borges solved both problems at once. First, they investigated using room-temperature ionic liquids (RTILs) developed in 2009 by European and Australian researchers. RTILs show low conductivity at room temperature but become less viscous and more conductive when heated.
Clay has high thermal stability, high sorption capacity, a large active surface area and high permeability, Reddy said, and is commonly used in muds for oil drilling, in modern construction, in medical applications and as a binder by iron and steel foundries.
After combining equal amounts of RTIL and naturally occurring Bentonite clay into a composite paste, the researchers sandwiched it between layers of reduced graphene oxide and two current collectors to form a supercapacitor. Tests and subsequent electron microscope images of the device showed no change in the materials after heating it to 200 degrees Celsius. In fact, Reddy said, there was very little change in the material up to 300 degrees Celsius.
"The ionic conductivity increases almost linearly until the material reaches 180 degrees, and then saturates at 200," he said.
Despite a slight drop in capacity observed in the initial charge/discharge cycles, the supercapacitors were stable through 10,000 test cycles. Both energy and power density improved by two orders of magnitude as the operating temperature increased from room temperature to 200 degrees Celsius, the researchers found.
The team took its discovery a step further and combined the RTIL/clay with a small amount of thermoplastic polyurethane to form a membrane sheet that can be cut into various shapes and sizes, which allows design flexibility for devices.
More information: www.nature.com/srep/2013/13090 … /full/srep02572.html
Journal information: Scientific Reports
Provided by Rice University