Genomic and computational tools provide window to distant past

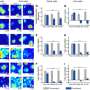
Out of the estimated 23,000 or more genes in the human genome, about 100 of them will differ—they will be present or not—between any two individuals. Genes lost or gained over time result from evolution and adaptation, as species respond through the years to their environment and other influences.
The availability of genomic sequences now allows scientists to study the presence or absence of whole genes among individuals and between species, and the impact of such changes for evolution.
Some individuals, for example, have a sharper sense of smell than others because they have more copies of olfactory receptor genes, which allow them to detect a wider range of odors. Others, especially those who live in societies with starchy diets, have more copies of the gene responsible for producing amylase, an enzyme in saliva that breaks down starch.
"There have been lots of changes, and we want to know which ones might have been involved in human adaptation," says Matthew Hahn, an associate professor of biology and informatics at Indiana University at Bloomington. "The comparison of whole genomes has revealed large and frequent changes in the size of gene families. Comparative genomic analyses allow us to identify large-scale patterns of change in gene families, and to make inferences regarding the role of natural selection in gene gain and loss."
Using computer models and available genomic data, Hahn studies the differences in genes among humans and other species, and compares them, in order to better understand the timeline of genetic changes and adaptation throughout our history. By developing computational and statistical tools to analyze whole genomes, Hahn and his team are learning new things about the evolution of gene regulation and gene families, human genomic history, and the evolution of phenotypically important genes.
"We can't go back in time, but we can use current species to get a pretty good estimate of what the ancestors looked like, and to get some ideas of what changes occurred and the order of these changes," he says.
The scientists are examining all the genes in the genome, and focusing on differences among species, such as chimpanzees and other primates compared to humans. "There's a 6 percent difference between humans and chimps in the genes they have," he says. "In the end, after 6 million years of being separate, we don't have exactly the same set of genes as chimps. How and when did those differences occur?"
Hahn is conducting his research under a National Science Foundation (NSF) Faculty Early Career Development (CAREER) award, which he received in 2009 as part of NSF's American Recovery and Reinvestment Act funding. The award supports junior faculty who exemplify the role of teacher-scholars through outstanding research, excellent education and the integration of education, and research within the context of the mission of their organization. He is receiving about $1 million over five years.
The work could have wide-ranging applications in diagnosing and treating diseases, since many illnesses and conditions arise from genetic mutations, including the duplication or loss of important genes.
"There is a lot of interest in trying to associate these changes to human diseases," Hahn says. "There are diseases that are caused when you lose or even gain a gene, not just affecting smell or the ability to digest starch. A lot of the genes that differ in copy number are genes involved in our immune response, and these are obvious candidates for the genetic changes underlying differences in disease susceptibility among individuals. By understanding normal variation in gene copy-number, we hope to be able to better recognize changes that may be detrimental to human health."
The researchers often start by examining the differences in the number of copies of different genes among individual humans.
"The 1,000 Genomes Project (an international research effort, launched in 2008, to establish the most detailed catalogue of human genetic variation) has allowed us to study the full genetic complement of genes in a wide variety of human populations, from all of the inhabited continents," he says. "We find differences between individuals within populations and among populations, largely recapitulating the known relationships among humans.
"But we also find population-specific changes in genes that have allowed us to adapt to our surroundings," he adds. "These changes have involved both the adaptive gain and adaptive loss of genes, and are associated with important phenotypic differences among individuals."
To understand the differences shared among all humans, and that distinguish us from our ancestors, the researchers then compare the full complement of genes to those of other primates, including chimpanzees, orangutans, macaques and marmosets.
"These comparisons, and similar ones to other new genomes that are being sequenced all the time, allow us to make strong inferences about what our common ancestral genome looked like, and, therefore, the changes that have occurred along the human lineage," he says.
Such genetic changes are highly likely to have been involved in human-specific adaptations, for example, humans' increased cranium size, according to Hahn.
"Having these genomic and computational tools gives us a window into the distant past that we otherwise would not have had," he says.
Provided by National Science Foundation