'Chemical architects' build materials with potential applications in drug delivery and gas storage
Home remodelers understand the concept of improving original foundations with more modern elements. Using this same approach—but with chemistry—researchers in the University of Pittsburgh's Kenneth P. Dietrich School of Arts and Sciences have designed a family of materials that could make drug delivery, gas storage, and gas transport more efficient and at a lower cost. The findings were reported in the latest issue of the Journal of the American Chemical Society (JACS).
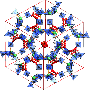
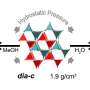
The recent work builds upon Pitt Associate Professor of Chemistry Nathaniel Rosi's earlier research published last year in Nature Communications detailing a new class of metal-organic frameworks—crystalline compounds consisting of metal vertices and organic linkers that form porous structures. Last year, Rosi and his team created one of the most porous materials known at the time by changing the size of the vertex (the metal cluster) rather than the length of the organic linkers. Now, in JACS, he and his team have extended those linkers, demonstrating a family of materials even more porous—a property necessary for more efficient gas storage.
"We like to think of ourselves as chemical architects," said Rosi, principal investigator of the project. "Our approach always starts with thinking about structure and, in particular, how we can design and manipulate structure. Here, we demonstrate one of the most porous families of metal-organic frameworks known."
Rosi likens his work to that of a builder remodeling a child's chair. As the child grows taller, the legs of the chair become too short. Because the owner likes the structure and integrity of the chair, the owner decides to lengthen its legs instead of purchasing a new one. This is what Rosi and his team have done with their frameworks: they have used one material as a structural blueprint and replaced another element (the organic linkers) to prepare more porous materials.
In addition to their utility for gas storage, these porous materials could be critical for low-cost industrial separations—when one molecule is separated from another batch of molecules for purification purposes. The petrochemical industry has numerous high-value (and high-cost) separations used to isolate important chemicals involved with oil refining. Some of these separations could benefit from the use of porous materials as filters, said Rosi. Likewise, he notes that the pore size for his class of materials would be particularly useful for separating nanoparticles. Porosity also can affect the efficiency of pharmaceutical delivery into the human body.
An important metric for evaluating the porosity of a material is its pore volume. In Rosi's demonstration, three of these materials have pore volumes exceeding 4 cubic centimeters per gram (cc/g). For perspective, only one other metal-organic framework has a pore volume above this amount, with most others having volumes below 3 cc/g.
"Pore volume is a measure of how empty or vacant a material is—how much space in the material isn't filled," said Rosi. "When the pore openings are large, and the pore volume is large, it opens up the possibility of using the material as a scaffold to precisely organize and position biomolecules or nanoparticles in space."
Rosi and his team are currently investigating high-porous and low-density materials to be used as scaffolds for organizing large molecules and nanoparticles into functional materials.
More information: The paper, "Stepwise Ligand Exchange for the Preparation of a Family of Mesoporous MOFs," was first published online May 20 in JACS.
Journal information: Journal of the American Chemical Society
Provided by University of Pittsburgh