Preventive detention for oxidizing agents: Role of oxidative stress needs to be re-evaluated
Oxidative stress is believed to cause a number of diseases. Up to now, it has been common practice to measure oxidative stress levels by determining the oxidation state of a small molecule called glutathione in cell extracts. Scientists from the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) have been the first to discover that cells under stress deposit their oxidized glutathione in a cellular waste repository. This protects cells from oxidative stress – and questions the validity of the conventional measuring method.
Cancer, Alzheimer's, arteriosclerosis– the list of diseases which have been linked to oxidative stress is long and even includes the very process of aging. Oxidative stress is caused by so-called reactive oxygen compounds, which include the notorious "free radicals". If a cell is exposed to more reactive oxygen compounds than it can instantly degrade, it is under oxidative stress. As a result, important components such as proteins, DNA and lipids are oxidized and thus get damaged.
To determine whether a cell is under oxidative stress, scientists often analyze the oxidation state of glutathione. Glutathione is a small molecule which gets oxidized to protect the cell from reactive oxygen compounds. In theory, the amount of oxidized glutathione should therefore indicate whether a cell is healthy or under oxidative stress. However, researchers in the team of Associate Professor (PD) Dr. Tobias Dick have demonstrated that this hypothesis, which is the basis of a large number of scientific studies, is deceptive.
"Up to now, it was necessary to destroy the cells in order to measure the amount of oxidized glutathione," Tobias Dick explains. "However, this means that any spatial resolution is lost." Therefore, virtually nothing was known about where exactly oxidized glutathione is found in the cells. Scientists have presumed that it remains in the cytoplasm, where it is formed.
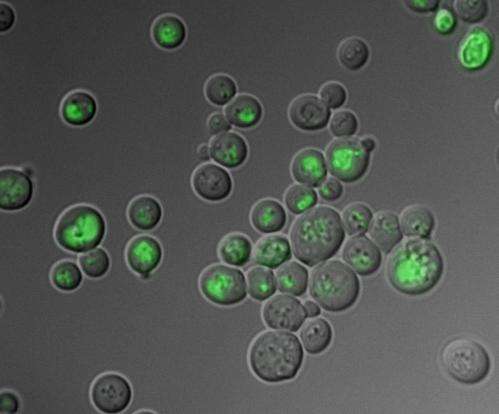
To find out more about the whereabouts of glutathione in the cell, Tobias Dick and co-workers developed biosensors which indicate the oxidation state of glutathione in intact cells by releasing light signals. In yeast cells, the researchers were able, for the first time, to follow the path of oxidized glutathione through the living cell in real time. They were surprised to find that, rather than remaining in the cytoplasm, it promptly gets locked up in a safe depot, the vacuole.
The cytoplasm, where all important cellular metabolic processes happen, is thus reliably protected from oxidative damage. Cells that would have been considered to be under oxidative stress using the conventional method appeared entirely healthy in their cytoplasm. Tobias Dick and his team could subsequently show that this is not only true for yeast cells but also for various mammalian cells and also for cancer cells.
These results mean that – contrary to previously held beliefs – the level of oxidative glutathione does not indicate whether or not a cell is under oxidative stress. "Therefore, it is important to re-evaluate prior studies that have established a link between oxidative stress and various diseases based on the conventional method."
More information: Bruce Morgan, Daria Ezeriņa, Theresa N.E. Amoako, Jan Riemer, Matthias Seedorf and Tobias P. Dick: Multiple glutathione disulfide removal pathways mediate cytosolic redox homeostasis. Nature Chemical Biology, 2012, DOI: 10.1038/NCHEMBIO.1142
Journal information: Nature Chemical Biology
Provided by Helmholtz Association of German Research Centres















