A second ascent of chemistry's Mt. Everest
In science's equivalent of ascending Mt. Everest, researchers are reporting success in one of the most difficult challenges in synthetic chemistry—a field in which scientists reproduce natural and other substances from jars of chemicals in a lab. The feat, reported in the Journal of the American Chemical Society, involved synthesis of a rare substance with promising in vitro anti-cancer effects found naturally in tiny amounts in a Chinese medicinal herb.
Samuel J. Danishefsky and Feng Peng explain that maoecrystal V occurs naturally in a plant growing in China's Yunnan province. Practitioners of Chinese traditional medicine have used the herb, called Isodon eriocalyx Hara, for hundreds of years. Scientists isolated maoecrystal V in 1994 and found that it has anti-cancer effects that warrant further examination. Large amounts would be needed to do so. However, the herb contains only small amounts of the material. In one experiment, it took more than 26 pounds of dried leaves to produce 0.00017637 oz. of maoecrystal V. In 2010, the first group of scientists succeeded in making maoecrystal V in a lab. Now, a second group has found a new route to the potential anti-cancer compound.
Danishefsky and Peng describe their approach as providing the basis for a synthesis that may help scientists develop a practical route to the substance for drug development and other research. It yielded a mixture of the two enantiomeric forms of maoecrystal V. They are pursuing several improvements, including a way to control the reactions to yield only a single enantiomer of the substance.
More information: "Total Synthesis of (±)-Maoecrystal V" J. Am. Chem. Soc., 2012, 134 (45), pp 18860–18867. DOI: 10.1021/ja309905j
Abstract
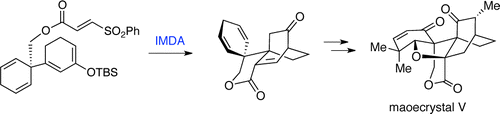
The total synthesis of racemic maoecrystal V has been accomplished. Key steps include an intramolecular Diels–Alder cyclization to rapidly construct the core system from simple starting materials and the creation of the A–C ring trans-fusion through intramolecular delivery of a hydrogen to the hindered β-face of the ring system.
Journal information: Journal of the American Chemical Society
Provided by American Chemical Society
















